Isolation of Foreign Material-Free Endothelial Progenitor Cells Using CD31 Aptamer and Therapeutic Application for Ischemic Injury
- PMID: 26148001
- PMCID: PMC4493074
- DOI: 10.1371/journal.pone.0131785
Isolation of Foreign Material-Free Endothelial Progenitor Cells Using CD31 Aptamer and Therapeutic Application for Ischemic Injury
Abstract
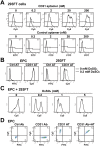
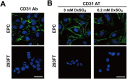
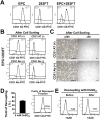
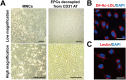
Endothelial progenitor cells (EPCs) can be isolated from human bone marrow or peripheral blood and reportedly contribute to neovascularization. Aptamers are 40-120-mer nucleotides that bind to a specific target molecule, as antibodies do. To utilize apatmers for isolation of EPCs, in the present study, we successfully generated aptamers that recognize human CD31, an endothelial cell marker. CD31 aptamers bound to human umbilical cord blood-derived EPCs and showed specific interaction with human CD31, but not with mouse CD31. However, CD31 aptamers showed non-specific interaction with CD31-negative 293FT cells and addition of polyanionic competitor dextran sulfate eliminated non-specific interaction without affecting cell viability. From the mixture of EPCs and 293FT cells, CD31 aptamers successfully isolated EPCs with 97.6% purity and 94.2% yield, comparable to those from antibody isolation. In addition, isolated EPCs were decoupled from CD31 aptamers with a brief treatment of high concentration dextran sulfate. EPCs isolated with CD31 aptamers and subsequently decoupled from CD31 aptamers were functional and enhanced the restoration of blood flow when transplanted into a murine hindlimb ischemia model. In this study, we demonstrated isolation of foreign material-free EPCs, which can be utilized as a universal protocol in preparation of cells for therapeutic transplantation.
Conflict of interest statement
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

