Borrelia burgdorferi RevA Significantly Affects Pathogenicity and Host Response in the Mouse Model of Lyme Disease
- PMID: 26150536
- PMCID: PMC4534657
- DOI: 10.1128/IAI.00530-15
Borrelia burgdorferi RevA Significantly Affects Pathogenicity and Host Response in the Mouse Model of Lyme Disease
Abstract
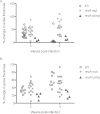
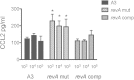
The Lyme disease spirochete, Borrelia burgdorferi, expresses RevA and numerous outer surface lipoproteins during mammalian infection. As an adhesin that promotes bacterial interaction with fibronectin, RevA is poised to interact with the extracellular matrix of the host. To further define the role(s) of RevA during mammalian infection, we created a mutant that is unable to produce RevA. The mutant was still infectious to mice, although it was significantly less well able to infect cardiac tissues. Complementation of the mutant with a wild-type revA gene restored heart infectivity to wild-type levels. Additionally, revA mutants led to increased evidence of arthritis, with increased fibrotic collagen deposition in tibiotarsal joints. The mutants also induced increased levels of the chemokine CCL2, a monocyte chemoattractant, in serum, and this increase was abolished in the complemented strain. Therefore, while revA is not absolutely essential for infection, deletion of revA had distinct effects on dissemination, arthritis severity, and host response.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures



Similar articles
-
Evaluation of RevA, a fibronectin-binding protein of Borrelia burgdorferi, as a potential vaccine candidate for lyme disease.Clin Vaccine Immunol. 2013 Jun;20(6):892-9. doi: 10.1128/CVI.00758-12. Epub 2013 Apr 17. Clin Vaccine Immunol. 2013. PMID: 23595502 Free PMC article.
-
Borrelia burgdorferi RevA antigen binds host fibronectin.Infect Immun. 2009 Jul;77(7):2802-12. doi: 10.1128/IAI.00227-09. Epub 2009 Apr 27. Infect Immun. 2009. PMID: 19398540 Free PMC article.
-
Borrelia burgdorferi RevA antigen is a surface-exposed outer membrane protein whose expression is regulated in response to environmental temperature and pH.Infect Immun. 2001 Sep;69(9):5286-93. doi: 10.1128/IAI.69.9.5286-5293.2001. Infect Immun. 2001. PMID: 11500397 Free PMC article.
-
A mouse model for Borrelia burgdorferi infection: approach to a vaccine against Lyme disease.Immunol Today. 1991 Jan;12(1):11-6. doi: 10.1016/0167-5699(91)90106-4. Immunol Today. 1991. PMID: 2015043 Review.
-
Outer surface protein polymorphisms linked to host-spirochete association in Lyme borreliae.Mol Microbiol. 2019 Apr;111(4):868-882. doi: 10.1111/mmi.14209. Epub 2019 Feb 27. Mol Microbiol. 2019. PMID: 30666741 Free PMC article. Review.
Cited by
-
Rhesus Brain Transcriptomic Landscape in an ex vivo Model of the Interaction of Live Borrelia Burgdorferi With Frontal Cortex Tissue Explants.Front Neurosci. 2019 Jun 28;13:651. doi: 10.3389/fnins.2019.00651. eCollection 2019. Front Neurosci. 2019. PMID: 31316336 Free PMC article.
-
mRNA transcript distribution bias between Borrelia burgdorferi bacteria and their outer membrane vesicles.FEMS Microbiol Lett. 2018 Jul 1;365(13):fny135. doi: 10.1093/femsle/fny135. FEMS Microbiol Lett. 2018. PMID: 29846577 Free PMC article.
-
Nanomechanical mechanisms of Lyme disease spirochete motility enhancement in extracellular matrix.Commun Biol. 2021 Mar 1;4(1):268. doi: 10.1038/s42003-021-01783-1. Commun Biol. 2021. PMID: 33649506 Free PMC article.
-
Past, present, and future of Lyme disease vaccines: antigen engineering approaches and mechanistic insights.Expert Rev Vaccines. 2022 Oct;21(10):1405-1417. doi: 10.1080/14760584.2022.2102484. Epub 2022 Jul 22. Expert Rev Vaccines. 2022. PMID: 35836340 Free PMC article. Review.
-
Borrelia burgdorferi Outer Surface Protein C Is Not the Sole Determinant of Dissemination in Mammals.Infect Immun. 2023 Apr 18;91(4):e0045622. doi: 10.1128/iai.00456-22. Epub 2023 Mar 7. Infect Immun. 2023. PMID: 36880751 Free PMC article.
References
-
- Franz JK, Fritze O, Rittig M, Keysser G, Priem S, Zacher J, Burmester GR, Krause A. 2001. Insights from a novel three-dimensional in vitro model of Lyme arthritis: standardized analysis of cellular and molecular interactions between Borrelia burgdorferi and synovial explants and fibroblasts. Arthritis Rheum 44:151–162. doi:10.1002/1529-0131(200101)44:1<151::AID-ANR19>3.0.CO;2-E. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

