Functional abnormalities in iPSC-derived cardiomyocytes generated from CPVT1 and CPVT2 patients carrying ryanodine or calsequestrin mutations
- PMID: 26153920
- PMCID: PMC4549051
- DOI: 10.1111/jcmm.12581
Functional abnormalities in iPSC-derived cardiomyocytes generated from CPVT1 and CPVT2 patients carrying ryanodine or calsequestrin mutations
Abstract
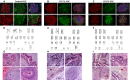
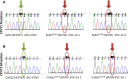
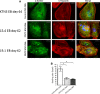
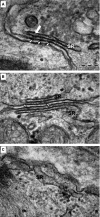
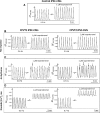
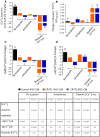
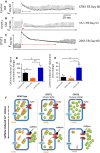
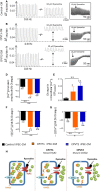
Catecholaminergic polymorphic ventricular tachycardia (CPVT) is an inherited arrhythmia characterized by syncope and sudden death occurring during exercise or acute emotion. CPVT is caused by abnormal intracellular Ca(2+) handling resulting from mutations in the RyR2 or CASQ2 genes. Because CASQ2 and RyR2 are involved in different aspects of the excitation-contraction coupling process, we hypothesized that these mutations are associated with different functional and intracellular Ca(²+) abnormalities. To test the hypothesis we generated induced Pluripotent Stem Cell-derived cardiomyocytes (iPSC-CM) from CPVT1 and CPVT2 patients carrying the RyR2(R420Q) and CASQ2(D307H) mutations, respectively, and investigated in CPVT1 and CPVT2 iPSC-CM (compared to control): (i) The ultrastructural features; (ii) the effects of isoproterenol, caffeine and ryanodine on the [Ca(2+) ]i transient characteristics. Our major findings were: (i) Ultrastructurally, CASQ2 and RyR2 mutated cardiomyocytes were less developed than control cardiomyocytes. (ii) While in control iPSC-CM isoproterenol caused positive inotropic and lusitropic effects, in the mutated cardiomyocytes isoproterenol was either ineffective, caused arrhythmias, or markedly increased diastolic [Ca(2+) ]i . Importantly, positive inotropic and lusitropic effects were not induced in mutated cardiomyocytes. (iii) The effects of caffeine and ryanodine in mutated cardiomyocytes differed from control cardiomyocytes. Our results show that iPSC-CM are useful for investigating the similarities/differences in the pathophysiological consequences of RyR2 versus CASQ2 mutations underlying CPVT1 and CPVT2 syndromes.
Keywords: Ca2+ transients; Induced pluripotent stem cells; arrhythmias; cardiomyocytes; catacholaminergic polymorphic ventricular tachycardia.
© 2015 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Figures

References
-
- Kontula K, Laitinen PJ, Lehtonen A, et al. Catecholaminergic polymorphic ventricular tachycardia: recent mechanistic insights. Cardiovasc Res. 2005;67:379–87. - PubMed
-
- Liu N, Priori SG. Disruption of calcium homeostasis and arrhythmogenesis induced by mutations in the cardiac ryanodine receptor and calsequestrin. Cardiovasc Res. 2008;77:293–301. - PubMed
-
- Itzhaki I, Maizels L, Huber I, et al. Modeling of catecholaminergic polymorphic ventricular tachycardia with patient-specific human-induced pluripotent stem cells. J Am Coll Cardiol. 2012;60:990–1000. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

