Single Chain Variable Fragments Produced in Escherichia coli against Heat-Labile and Heat-Stable Toxins from Enterotoxigenic E. coli
- PMID: 26154103
- PMCID: PMC4496030
- DOI: 10.1371/journal.pone.0131484
Single Chain Variable Fragments Produced in Escherichia coli against Heat-Labile and Heat-Stable Toxins from Enterotoxigenic E. coli
Abstract
Background: Diarrhea is a prevalent pathological condition frequently associated to the colonization of the small intestine by enterotoxigenic Escherichia coli (ETEC) strains, known to be endemic in developing countries. These strains can produce two enterotoxins associated with the manifestation of clinical symptoms that can be used to detect these pathogens. Although several detection tests have been developed, minimally equipped laboratories are still in need of simple and cost-effective methods. With the aim to contribute to the development of such diagnostic approaches, we describe here two mouse hybridoma-derived single chain fragment variable (scFv) that were produced in E. coli against enterotoxins of ETEC strains.
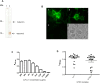
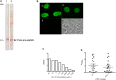
Methods and findings: Recombinant scFv were developed against ETEC heat-labile toxin (LT) and heat-stable toxin (ST), from previously isolated hybridoma clones. This work reports their design, construction, molecular and functional characterization against LT and ST toxins. Both antibody fragments were able to recognize the cell-interacting toxins by immunofluorescence, the purified toxins by ELISA and also LT-, ST- and LT/ST-producing ETEC strains.
Conclusion: The developed recombinant scFvs against LT and ST constitute promising starting point for simple and cost-effective ETEC diagnosis.
Conflict of interest statement
Figures


References
-
- World Health Organization. Diarrhoea disease. 2013 Available: http://www.who.int/mediacentre/factsheets/fs330/en/index.html.
-
- Hill DR, Beeching NJ. Travelers’ diarrhea. Curr Opin Infect Dis. 2010; 23:481–487. . - PubMed
-
- Sack DA. Management of acute infectious diarrhea. Compr Ther. 1980; 6:50–56. . - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

