High Sustained Virologic Response to Daclatasvir Plus Asunaprevir in Elderly and Cirrhotic Patients with Hepatitis C Virus Genotype 1b Without Baseline NS5A Polymorphisms
- PMID: 26155891
- PMCID: PMC4522028
- DOI: 10.1007/s12325-015-0221-5
High Sustained Virologic Response to Daclatasvir Plus Asunaprevir in Elderly and Cirrhotic Patients with Hepatitis C Virus Genotype 1b Without Baseline NS5A Polymorphisms
Abstract
Introduction: Oral daclatasvir (DCV; pangenotypic NS5A inhibitor) plus asunaprevir (ASV; NS3 protease inhibitor) is approved in Japan and Korea for treatment of chronic hepatitis C virus (HCV) genotype 1. Response to DCV + ASV is affected by DCV resistance-associated polymorphisms (RAPs) in HCV NS5A. The prevalence and influence of these RAPs on 12-week sustained virologic response (SVR12) to DCV + ASV was evaluated in Asian and non-Asian patients.
Methods: Data were pooled from 5 national and international studies of patients with HCV genotype 1b (GT-1b) receiving DCV + ASV at their recommended doses. Baseline NS5A RAPs and their effect on SVR12 were assessed overall, in older (≥65 years) patients, patients with cirrhosis, and in patients stratified by baseline HCV RNA or prior treatment experience with interferon-based therapy.
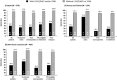
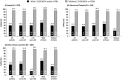
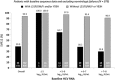
Results: Baseline NS5A sequences were available from 988 patients (374 Japanese; 125 Korean/Taiwanese; 489 from non-Asian countries), 979 of whom were assessed for SVR12. Pretreatment NS5A-L31F/I/M/V and/or NS5A-Y93H polymorphisms were present in 18% of Japanese and 12-13% of non-Japanese patients; these RAPs reduced SVR12 by 54.9% overall (93.9% [787/838] SVR12 when absent, 39.0% [55/141] SVR12 when present), with comparable reductions observed in Asians and non-Asians and across all categories of treatment experience, age, and cirrhosis. RAP-associated SVR12 rates declined with increasing baseline HCV RNA (SVR12 with RAPs: 64.7% [11/17] at 5-6 log10 IU/mL, 29.8% [14/47] at 7-8 log10). Without baseline RAPs, very high SVR12 rates (92-100%) were observed in older patients and patients with cirrhosis irrespective of national origin, with similarly high rates observed among treatment-naïve and interferon-experienced patients and those with high baseline HCV RNA.
Conclusions: Following DCV + ASV treatment, the SVR12 rates in GT-1b patients without baseline NS5A-L31F/I/M/V and/or NS5A-Y93H polymorphisms were very high (approximately 90-100%), irrespective of age, cirrhosis, prior interferon treatment, or baseline HCV RNA.
Funding: Bristol-Myers Squibb.
Figures


References
-
- World Health Organization. Hepatitis C Key Facts. WHO factsheet No 164. 2014. http://www.who.int/mediacentre/factsheets/fs164/en/. Accessed Apr 2015.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

