Treatment of chronic migraine with transcutaneous stimulation of the auricular branch of the vagal nerve (auricular t-VNS): a randomized, monocentric clinical trial
- PMID: 26156114
- PMCID: PMC4496420
- DOI: 10.1186/s10194-015-0543-3
Treatment of chronic migraine with transcutaneous stimulation of the auricular branch of the vagal nerve (auricular t-VNS): a randomized, monocentric clinical trial
Abstract
Background: Aim of the study was assessment of efficacy and safety of transcutaneous stimulation of the auricular branch of the vagal nerve (t-VNS) in the treatment of chronic migraine.
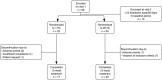
Methods: A monocentric, randomized, controlled, double-blind study was conducted. After one month of baseline, chronic migraine patients were randomized to receive 25 Hz or 1 Hz stimulation of the sensory vagal area at the left ear by a handhold battery driven stimulator for 4 h/day during 3 months. Headache days per 28 days were compared between baseline and the last month of treatment and the number of days with acute medication was recorded The Headache Impact Test (HIT-6) and the Migraine Disability Assessment (MIDAS) questionnaires were used to assess headache-related disability.
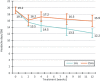
Results: Of 46 randomized patients, 40 finished the study (per protocol). In the per protocol analysis, patients in the 1 Hz group had a significantly larger reduction in headache days per 28 days than patients in the 25 Hz group (-7.0 ± 4.6 vs. -3.3 ± 5.4 days, p = 0.035). 29.4 % of the patients in the 1 Hz group had a ≥50 % reduction in headache days vs. 13.3 % in the 25 Hz group. HIT-6 and MIDAS scores were significantly improved in both groups, without group differences. There were no serious treatment-related adverse events.
Conclusion: Treatment of chronic migraine by t-VNS at 1 Hz was safe and effective. The mean reduction of headache days after 12 weeks of treatment exceeded that reported for other nerve stimulating procedures.
Figures


Similar articles
-
Transcutaneous Vagus Nerve Stimulation (tVNS) for Treatment of Drug-Resistant Epilepsy: A Randomized, Double-Blind Clinical Trial (cMPsE02).Brain Stimul. 2016 May-Jun;9(3):356-363. doi: 10.1016/j.brs.2015.11.003. Epub 2016 Jan 20. Brain Stimul. 2016. PMID: 27033012 Clinical Trial.
-
Safety and efficacy of peripheral nerve stimulation of the occipital nerves for the management of chronic migraine: long-term results from a randomized, multicenter, double-blinded, controlled study.Cephalalgia. 2015 Apr;35(4):344-58. doi: 10.1177/0333102414543331. Epub 2014 Jul 30. Cephalalgia. 2015. PMID: 25078718 Clinical Trial.
-
Remote Electrical Neuromodulation (REN) Relieves Acute Migraine: A Randomized, Double-Blind, Placebo-Controlled, Multicenter Trial.Headache. 2019 Sep;59(8):1240-1252. doi: 10.1111/head.13551. Epub 2019 May 9. Headache. 2019. PMID: 31074005 Free PMC article. Clinical Trial.
-
Management of auricular transcutaneous neuromodulation and electro-acupuncture of the vagus nerve for chronic migraine: a systematic review.Front Neurosci. 2023 Jun 15;17:1151892. doi: 10.3389/fnins.2023.1151892. eCollection 2023. Front Neurosci. 2023. PMID: 37397439 Free PMC article. Review.
-
Lessons learned from transcutaneous vagus nerve stimulation (tVNS).Epilepsy Res. 2019 Jul;153:83-84. doi: 10.1016/j.eplepsyres.2019.02.015. Epub 2019 Feb 26. Epilepsy Res. 2019. PMID: 30952581 Review.
Cited by
-
Transcutaneous auricular vagus nerve stimulation at 1 Hz modulates locus coeruleus activity and resting state functional connectivity in patients with migraine: An fMRI study.Neuroimage Clin. 2019;24:101971. doi: 10.1016/j.nicl.2019.101971. Epub 2019 Aug 5. Neuroimage Clin. 2019. PMID: 31648171 Free PMC article. Clinical Trial.
-
The Use of Electrical Nerve Stimulation to Treat Migraines: A Systematic Review.Cureus. 2021 Aug 30;13(8):e17554. doi: 10.7759/cureus.17554. eCollection 2021 Aug. Cureus. 2021. PMID: 34646611 Free PMC article. Review.
-
Auricular vagus nerve stimulation: a new option to treat inflammation in COVID-19?Rev Assoc Med Bras (1992). 2023 Jun 2;69(6):e20230345. doi: 10.1590/1806-9282.20230345. eCollection 2023. Rev Assoc Med Bras (1992). 2023. PMID: 37283364 Free PMC article. No abstract available.
-
Auricular Vagal Neuromodulation and its Application in Patients with Heart Failure and Reduced Ejection Fraction.Arq Bras Cardiol. 2023 May 15;120(5):e20220581. doi: 10.36660/abc.20220581. eCollection 2023. Arq Bras Cardiol. 2023. PMID: 37194830 Free PMC article. Clinical Trial. English, Portuguese.
-
Noninvasive vagus nerve stimulation for migraine: a systematic review and meta-analysis of randomized controlled trials.Front Neurol. 2023 May 11;14:1190062. doi: 10.3389/fneur.2023.1190062. eCollection 2023. Front Neurol. 2023. PMID: 37251233 Free PMC article. Review.
References
-
- Diener HC, Dodick DW, Aurora SK, Turkel CC, DeGryse RE, Lipton RB, Silberstein SD, Brin MF. OnabotulinumtoxinA for treatment of chronic migraine: results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 2 trial. Cephalalgia. 2010;30:804–814. doi: 10.1177/0333102410364677. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

