Comparison of genetically encoded calcium indicators for monitoring action potentials in mammalian brain by two-photon excitation fluorescence microscopy
- PMID: 26158004
- PMCID: PMC4478877
- DOI: 10.1117/1.NPh.2.2.021014
Comparison of genetically encoded calcium indicators for monitoring action potentials in mammalian brain by two-photon excitation fluorescence microscopy
Abstract
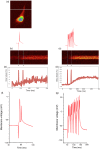
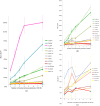
Imaging calcium transients associated with neuronal activity has yielded important insights into neural physiology. Genetically encoded calcium indicators (GECIs) offer conspicuous potential advantages for this purpose, including exquisite targeting. While the catalogue of available GECIs is steadily growing, many newly developed sensors that appear promising in vitro or in model cells appear to be less useful when expressed in mammalian neurons. We have, therefore, evaluated the performance of GECIs from two of the most promising families of sensors, G-CaMPs [Nat. Biotechnol.2(2), 137-141 (2001)] and GECOs [Science2(2), 1888-1891 (2011)], for monitoring action potentials in rat brain. Specifically, we used two-photon excitation fluorescence microscopy to compare calcium transients detected by G-CaMP3; GCaMP6f; G-CaMP7; Green-GECO1.0, 1.1 and 1.2; Blue-GECO; Red-GECO; Rex-GECO0.9; Rex-GECO1; Carmine-GECO; Orange-GECO; and Yellow-GECO1s. After optimizing excitation wavelengths, we monitored fluorescence signals associated with increasing numbers of action potentials evoked by current injection in CA1 pyramidal neurons in rat organotypic hippocampal slices. Some GECIs, particularly Green-GECO1.2, GCaMP6f, and G-CaMP7, were able to detect single action potentials with high reliability. By virtue of greatest sensitivity and fast kinetics, G-CaMP7 may be the best currently available GECI for monitoring calcium transients in mammalian neurons.
Keywords: G-CaMP; GECO; calcium transients; genetically encoded calcium indicators; organotypic hippocampal slice; two-photon microscopy.
Figures
Similar articles
-
Blue-shifted genetically encoded Ca2+ indicator with enhanced two-photon absorption.Neurophotonics. 2024 Apr;11(2):024207. doi: 10.1117/1.NPh.11.2.024207. Epub 2024 Apr 4. Neurophotonics. 2024. PMID: 38577628 Free PMC article.
-
A blue-shifted genetically encoded Ca2+ indicator with enhanced two-photon absorption.bioRxiv [Preprint]. 2023 Oct 17:2023.10.12.562058. doi: 10.1101/2023.10.12.562058. bioRxiv. 2023. Update in: Neurophotonics. 2024 Apr;11(2):024207. doi: 10.1117/1.NPh.11.2.024207. PMID: 37905143 Free PMC article. Updated. Preprint.
-
Quantitative comparison of novel GCaMP-type genetically encoded Ca(2+) indicators in mammalian neurons.Front Cell Neurosci. 2012 Oct 8;6:41. doi: 10.3389/fncel.2012.00041. eCollection 2012. Front Cell Neurosci. 2012. PMID: 23060748 Free PMC article.
-
Imaging neuronal activity with genetically encoded calcium indicators.Cold Spring Harb Protoc. 2012 Jun 1;2012(6):647-56. doi: 10.1101/pdb.top069609. Cold Spring Harb Protoc. 2012. PMID: 22661439 Review.
-
Reporting neural activity with genetically encoded calcium indicators.Brain Cell Biol. 2008 Aug;36(1-4):69-86. doi: 10.1007/s11068-008-9029-4. Epub 2008 Oct 22. Brain Cell Biol. 2008. PMID: 18941901 Free PMC article. Review.
Cited by
-
Insights Into Spinal Dorsal Horn Circuit Function and Dysfunction Using Optical Approaches.Front Neural Circuits. 2020 Jun 12;14:31. doi: 10.3389/fncir.2020.00031. eCollection 2020. Front Neural Circuits. 2020. PMID: 32595458 Free PMC article. Review.
-
Lysosome and plasma membrane Piezo channels of Trypanosoma cruzi are essential for proliferation, differentiation and infectivity.PLoS Pathog. 2025 Apr 23;21(4):e1013105. doi: 10.1371/journal.ppat.1013105. eCollection 2025 Apr. PLoS Pathog. 2025. PMID: 40267157 Free PMC article.
-
Design and mechanistic insight into ultrafast calcium indicators for monitoring intracellular calcium dynamics.Sci Rep. 2016 Dec 6;6:38276. doi: 10.1038/srep38276. Sci Rep. 2016. PMID: 27922063 Free PMC article.
-
Neuronal Activity Reporters as Drug Screening Platforms.Micromachines (Basel). 2022 Sep 9;13(9):1500. doi: 10.3390/mi13091500. Micromachines (Basel). 2022. PMID: 36144123 Free PMC article. Review.
-
Probing Single Synapses via the Photolytic Release of Neurotransmitters.Front Synaptic Neurosci. 2019 Jul 12;11:19. doi: 10.3389/fnsyn.2019.00019. eCollection 2019. Front Synaptic Neurosci. 2019. PMID: 31354469 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

