Robust hematopoietic progenitor cell commitment in the presence of a conflicting cue
- PMID: 26159733
- PMCID: PMC4541040
- DOI: 10.1242/jcs.158436
Robust hematopoietic progenitor cell commitment in the presence of a conflicting cue
Erratum in
-
Robust hematopoietic progenitor cell commitment in the presence of a conflicting cue.J Cell Sci. 2015 Nov 1;128(21):4024. doi: 10.1242/jcs.180653. J Cell Sci. 2015. PMID: 26527202 Free PMC article. No abstract available.
Abstract
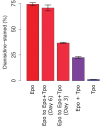
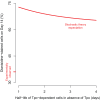
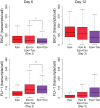
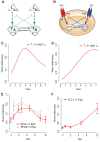
Hematopoietic lineage commitment is regulated by cytokines and master transcription factors, but it remains unclear how a progenitor cell chooses a lineage in the face of conflicting cues. Through transcript counting in megakaryocyte-erythroid progenitors undergoing erythropoiesis, we show that the expression levels of the pro-erythropoiesis transcription factor EKLF (also known as KLF1) and receptor EpoR are inversely correlated with their pro-megakaryopoiesis counterparts, FLI-1 and TpoR (also known as MPL). Notably, as progenitors commit to the erythrocyte lineage, EpoR is upregulated and TpoR is strongly downregulated, thus boosting the potency of the pro-erythropoiesis cue erythropoietin and effectively eliminating the activity of the pro-megakaryopoiesis cue thrombopoietin. Based on these findings, we propose a new model for exclusive decision making that explicitly incorporates signals from extrinsic cues, and we experimentally confirm a model prediction of temporal changes in transcript noise levels in committing progenitors. Our study suggests that lineage-specific receptor levels can modulate potencies of cues to achieve robust commitment decisions.
Keywords: Conflicting cue; Hematopoiesis; Lineage commitment.
© 2015. Published by The Company of Biologists Ltd.
Conflict of interest statement
The authors declare no competing or financial interests.
Figures


Similar articles
-
Novel role for EKLF in megakaryocyte lineage commitment.Blood. 2007 Dec 1;110(12):3871-80. doi: 10.1182/blood-2007-03-082065. Epub 2007 Aug 22. Blood. 2007. PMID: 17715392 Free PMC article.
-
Inducible Fli-1 gene deletion in adult mice modifies several myeloid lineage commitment decisions and accelerates proliferation arrest and terminal erythrocytic differentiation.Blood. 2010 Dec 2;116(23):4795-805. doi: 10.1182/blood-2010-02-270405. Epub 2010 Aug 23. Blood. 2010. PMID: 20733157
-
Downregulation of signal transducer and activator of transcription 5 (STAT5) in CD34+ cells promotes megakaryocytic development, whereas activation of STAT5 drives erythropoiesis.Stem Cells. 2008 Jul;26(7):1732-42. doi: 10.1634/stemcells.2007-0899. Epub 2008 Apr 24. Stem Cells. 2008. PMID: 18436865
-
Megakaryocyte development and platelet production.Br J Haematol. 2006 Sep;134(5):453-66. doi: 10.1111/j.1365-2141.2006.06215.x. Br J Haematol. 2006. PMID: 16856888 Review.
-
The multifunctional role of EKLF/KLF1 during erythropoiesis.Blood. 2011 Aug 25;118(8):2044-54. doi: 10.1182/blood-2011-03-331371. Epub 2011 May 25. Blood. 2011. PMID: 21613252 Free PMC article. Review.
Cited by
-
Trajectory-based energy landscapes of gene regulatory networks.Biophys J. 2021 Feb 16;120(4):687-698. doi: 10.1016/j.bpj.2020.11.2279. Epub 2021 Jan 14. Biophys J. 2021. PMID: 33453275 Free PMC article.
-
Variable cellular decision-making behavior in a constant synthetic network topology.BMC Bioinformatics. 2019 May 14;20(1):237. doi: 10.1186/s12859-019-2866-6. BMC Bioinformatics. 2019. PMID: 31088350 Free PMC article.
-
Towards a Quantitative Understanding of Cell Identity.Trends Cell Biol. 2018 Dec;28(12):1030-1048. doi: 10.1016/j.tcb.2018.09.002. Epub 2018 Oct 8. Trends Cell Biol. 2018. PMID: 30309735 Free PMC article. Review.
References
-
- Bastian L. S., Kwiatkowski B. A., Breininger J., Danner S. and Roth G. (1999). Regulation of the megakaryocytic glycoprotein IX promoter by the oncogenic Ets transcription factor Fli-1. Blood 93, 2637-2644. - PubMed
-
- Borg J., Papadopoulos P., Georgitsi M., Gutiérrez L., Grech G., Fanis P., Phylactides M., Verkerk A. J. M. H., van der Spek P. J., Scerri C. A. et al. (2010). Haploinsufficiency for the erythroid transcription factor KLF1 causes hereditary persistence of fetal hemoglobin. Nat. Genet. 42, 801-805. 10.1038/ng.630 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

