Persistent Antigen and Prolonged AKT-mTORC1 Activation Underlie Memory CD8 T Cell Impairment in the Absence of CD4 T Cells
- PMID: 26163589
- PMCID: PMC4530017
- DOI: 10.4049/jimmunol.1500451
Persistent Antigen and Prolonged AKT-mTORC1 Activation Underlie Memory CD8 T Cell Impairment in the Absence of CD4 T Cells
Abstract
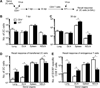
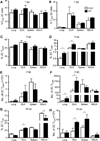
Recall responses by memory CD8 T cells are impaired in the absence of CD4 T cells. Although several mechanisms have been proposed, the molecular basis is still largely unknown. Using a local influenza virus infection in the respiratory tract and the lung of CD4(-/-) mice, we show that memory CD8 T cell impairment is limited to the lungs and the lung-draining lymph nodes, where viral Ags are unusually persistent and abundant in these mice. Persistent Ag exposure results in prolonged activation of the AKT-mTORC1 pathway in Ag-specific CD8 T cells, favoring their development into effector memory T cells at the expense of central memory T cells, and inhibition of mTORC1 by rapamycin largely corrects the impairment by promoting central memory T cell development. The findings suggest that the prolonged AKT-mTORC1 activation driven by persistent Ag is a critical mechanism underlying the impaired memory CD8 T cell development and responses in the absence of CD4 T cells.
Copyright © 2015 by The American Association of Immunologists, Inc.
Conflict of interest statement
The authors have no financial conflict of interest.
Figures

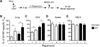
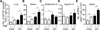

Similar articles
-
mTORC1 and mTORC2 selectively regulate CD8⁺ T cell differentiation.J Clin Invest. 2015 May;125(5):2090-108. doi: 10.1172/JCI77746. Epub 2015 Apr 20. J Clin Invest. 2015. PMID: 25893604 Free PMC article.
-
Aberrant CD8+ T-cell responses and memory differentiation upon viral infection of an ataxia-telangiectasia mouse model driven by hyper-activated Akt and mTORC1 signaling.Am J Pathol. 2011 Jun;178(6):2740-2751. doi: 10.1016/j.ajpath.2011.02.022. Am J Pathol. 2011. PMID: 21641396 Free PMC article.
-
Quantitative analysis of the influenza virus-specific CD4+ T cell memory in the absence of B cells and Ig.J Immunol. 1996 Oct 1;157(7):2947-52. J Immunol. 1996. PMID: 8816401
-
mTORC1 regulates CD8+ T-cell glucose metabolism and function independently of PI3K and PKB.Biochem Soc Trans. 2013 Apr;41(2):681-6. doi: 10.1042/BST20120359. Biochem Soc Trans. 2013. PMID: 23514176 Review.
-
Control of innate immunity by memory CD4 T cells.Adv Exp Med Biol. 2011;780:57-68. doi: 10.1007/978-1-4419-5632-3_6. Adv Exp Med Biol. 2011. PMID: 21842365 Free PMC article. Review.
Cited by
-
Differential Kinase Activation in Peripheral Blood Mononuclear Cells from Non-Small-Cell Lung Cancer Patients Treated with Nivolumab.Cancers (Basel). 2019 May 31;11(6):762. doi: 10.3390/cancers11060762. Cancers (Basel). 2019. PMID: 31159331 Free PMC article.
-
iCa2+ Flux, ROS and IL-10 Determines Cytotoxic, and Suppressor T Cell Functions in Chronic Human Viral Infections.Front Immunol. 2020 Mar 6;11:83. doi: 10.3389/fimmu.2020.00083. eCollection 2020. Front Immunol. 2020. PMID: 32210950 Free PMC article.
-
Cascade amplification of therapeutic payloads for cancer immunotherapy.Commun Biol. 2025 Aug 25;8(1):1277. doi: 10.1038/s42003-025-08735-z. Commun Biol. 2025. PMID: 40851032 Free PMC article.
-
Tissue-resident memory T cells and their biological characteristics in the recurrence of inflammatory skin disorders.Cell Mol Immunol. 2020 Jan;17(1):64-75. doi: 10.1038/s41423-019-0291-4. Epub 2019 Oct 8. Cell Mol Immunol. 2020. PMID: 31595056 Free PMC article. Review.
-
Helpless Priming Sends CD8+ T Cells on the Road to Exhaustion.Front Immunol. 2020 Oct 6;11:592569. doi: 10.3389/fimmu.2020.592569. eCollection 2020. Front Immunol. 2020. PMID: 33123174 Free PMC article.
References
-
- Shedlock DJ, Shen H. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science. 2003;300:337–339. - PubMed
-
- Janssen EM, Lemmens EE, Wolfe T, Christen U, von Herrath MG, Schoenberger SP. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature. 2003;421:852–856. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

