The CPCFC cuticular protein family: Anatomical and cuticular locations in Anopheles gambiae and distribution throughout Pancrustacea
- PMID: 26164413
- PMCID: PMC4628598
- DOI: 10.1016/j.ibmb.2015.07.002
The CPCFC cuticular protein family: Anatomical and cuticular locations in Anopheles gambiae and distribution throughout Pancrustacea
Abstract
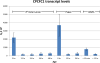
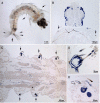
Arthropod cuticles have, in addition to chitin, many structural proteins belonging to diverse families. Information is sparse about how these different cuticular proteins contribute to the cuticle. Most cuticular proteins lack cysteine with the exception of two families (CPAP1 and CPAP3), recently described, and the one other that we now report on that has a motif of 16 amino acids first identified in a protein, Bc-NCP1, from the cuticle of nymphs of the cockroach, Blaberus craniifer (Jensen et al., 1997). This motif turns out to be present as two or three copies in one or two proteins in species from many orders of Hexapoda. We have named the family of cuticular proteins with this motif CPCFC, based on its unique feature of having two cysteines interrupted by five amino acids (C-X(5)-C). Analysis of the single member of the family in Anopheles gambiae (AgamCPCFC1) revealed that its mRNA is most abundant immediately following ecdysis in larvae, pupae and adults. The mRNA is localized primarily in epidermis that secretes hard cuticle, sclerites, setae, head capsules, appendages and spermatheca. EM immunolocalization revealed the presence of the protein, generally in endocuticle of legs and antennae. A phylogenetic analysis found proteins bearing this motif in 14 orders of Hexapoda, but not in some species for which there are complete genomic data. Proteins were much longer in Coleoptera and Diptera than in other orders. In contrast to the 1 and occasionally 2 copies in other species, a dragonfly, Ladona fulva, has at least 14 genes coding for family members. CPCFC proteins were present in four classes of Crustacea with 5 repeats in one species, and motifs that ended C-X(7)-C in Malacostraca. They were not detected, except as obvious contaminants, in any other arthropod subphyla or in any other phylum. The conservation of CPCFC proteins throughout the Pancrustacea and the small number of copies in individual species indicate that, when present, these proteins are serving important functions worthy of further study.
Keywords: Arthropod phylogeny; Cuticle; EM immunolocalization; In situ hybridization; RT-qPCR.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures






Similar articles
-
Immunolocalization of cuticular proteins in Johnston's organ and the corneal lens of Anopheles gambiae.Arthropod Struct Dev. 2016 Nov;45(6):519-535. doi: 10.1016/j.asd.2016.10.006. Epub 2016 Nov 4. Arthropod Struct Dev. 2016. PMID: 27744002 Free PMC article.
-
CPF and CPFL, two related gene families encoding cuticular proteins of Anopheles gambiae and other insects.Insect Biochem Mol Biol. 2007 Jul;37(7):675-88. doi: 10.1016/j.ibmb.2007.03.011. Epub 2007 Mar 31. Insect Biochem Mol Biol. 2007. PMID: 17550824
-
CutProtFam-Pred: detection and classification of putative structural cuticular proteins from sequence alone, based on profile hidden Markov models.Insect Biochem Mol Biol. 2014 Sep;52:51-9. doi: 10.1016/j.ibmb.2014.06.004. Epub 2014 Jun 27. Insect Biochem Mol Biol. 2014. PMID: 24978609 Free PMC article.
-
Exoskeletons across the Pancrustacea: Comparative Morphology, Physiology, Biochemistry and Genetics.Integr Comp Biol. 2015 Nov;55(5):771-91. doi: 10.1093/icb/icv080. Epub 2015 Jun 30. Integr Comp Biol. 2015. PMID: 26136336 Review.
-
Structural cuticular proteins from arthropods: annotation, nomenclature, and sequence characteristics in the genomics era.Insect Biochem Mol Biol. 2010 Mar;40(3):189-204. doi: 10.1016/j.ibmb.2010.02.001. Epub 2010 Feb 18. Insect Biochem Mol Biol. 2010. PMID: 20171281 Free PMC article. Review.
Cited by
-
Proteomics reveals localization of cuticular proteins in Anopheles gambiae.Insect Biochem Mol Biol. 2019 Jan;104:91-105. doi: 10.1016/j.ibmb.2018.09.011. Epub 2018 Sep 29. Insect Biochem Mol Biol. 2019. PMID: 30278207 Free PMC article.
-
Unraveling the Role of Cuticular Protein 3-like (HvCP3L) in the Chitin Pathway through RNAi and Methoxyfenozide Stress Response in Heortia vitessoides Moore.Insects. 2024 May 16;15(5):362. doi: 10.3390/insects15050362. Insects. 2024. PMID: 38786918 Free PMC article.
-
Immunolocalization of cuticular proteins in Johnston's organ and the corneal lens of Anopheles gambiae.Arthropod Struct Dev. 2016 Nov;45(6):519-535. doi: 10.1016/j.asd.2016.10.006. Epub 2016 Nov 4. Arthropod Struct Dev. 2016. PMID: 27744002 Free PMC article.
-
Culex pipiens pallens cuticular protein CPLCG5 participates in pyrethroid resistance by forming a rigid matrix.Parasit Vectors. 2018 Jan 4;11(1):6. doi: 10.1186/s13071-017-2567-9. Parasit Vectors. 2018. PMID: 29301564 Free PMC article.
-
A Novel miRNA-hlo-miR-2-Serves as a Regulatory Factor That Controls Molting Events by Targeting CPR1 in Haemaphysalis longicornis Nymphs.Front Microbiol. 2020 May 29;11:1098. doi: 10.3389/fmicb.2020.01098. eCollection 2020. Front Microbiol. 2020. PMID: 32547523 Free PMC article.
References
-
- Bustin SA, Benes V, Garson J, Hellemans J, Huggett J, Kubista M, Mueller R, Nolan T, Pfaffl MW, Shipley G, Wittwer CT, Schjerling P, Day PJ, Abreu M, Aguado B, Beaulieu JF, Beckers A, Bogaert S, Browne JA, Carrasco-Ramiro F, Ceelen L, Ciborowski K, Cornillie P, Coulon S, Cuypers A, De Brouwer S, De Ceuninck L, De Craene J, De Naeyer H, De Spiegelaere W, Deckers K, Dheedene A, Durinck K, Ferreira-Teixeira M, Fieuw A, Gallup JM, Gonzalo-Flores S, Goossens K, Heindryckx F, Herring E, Hoenicka H, Icardi L, Jaggi R, Javad F, Karampelias M, Kibenge F, Kibenge M, Kumps C, Lambertz I, Lammens T, Markey A, Messiaen P, Mets E, Morais S, Mudarra-Rubio A, Nakiwala J, Nelis H, Olsvik PA, Perez-Novo C, Plusquin M, Remans T, Rihani A, Rodrigues-Santos P, Rondou P, Sanders R, Schmidt-Bleek K, Skovgaard K, Smeets K, Tabera L, Toegel S, Van Acker T, Van den Broeck W, Van der Meulen J, Van Gele M, Van Peer G, Van Poucke M, Van Roy N, Vergult S, Wauman J, Tshuikina-Wiklander M, Willems E, Zaccara S, Zeka F, Vandesompele J. The need for transparency and good practices in the qPCR literature. Nat. Methods. 2013;10:1063–1067. - PubMed
-
- Colbourne JK, Pfrender ME, Gilbert D, Thomas WK, Tucker A, Oakley TH, Tokishita S, Aerts A, Arnold GJ, Basu MK, Bauer DJ, Caceres CE, Carmel L, Casola C, Choi JH, Detter JC, Dong Q, Dusheyko S, Eads BD, Frohlich T, Geiler-Samerotte KA, Gerlach D, Hatcher P, Jogdeo S, Krijgsveld J, Kriventseva EV, Kultz D, Laforsch C, Lindquist E, Lopez J, Manak JR, Muller J, Pangilinan J, Patwardhan RP, Pitluck S, Pritham EJ, Rechtsteiner A, Rho M, Rogozin IB, Sakarya O, Salamov A, Schaack S, Shapiro H, Shiga Y, Skalitzky C, Smith Z, Souvorov A, Sung W, Tang Z, Tsuchiya D, Tu H, Vos H, Wang M, Wolf YI, Yamagata H, Yamada T, Ye Y, Shaw JR, Andrews J, Crease TJ, Tang H, Lucas SM, Robertson HM, Bork P, Koonin EV, Zdobnov EM, Grigoriev IV, Lynch M, Boore JL. The ecoresponsive genome of Daphnia pulex. Science. 2011;311:555–561. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

