Multidisciplinary clinical management of paroxysmal nocturnal hemoglobinuria
- PMID: 26171279
- PMCID: PMC4497492
Multidisciplinary clinical management of paroxysmal nocturnal hemoglobinuria
Abstract
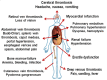
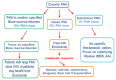
Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, acquired disease caused by clonal expansion of one or more hematopoietic stem cell (HSC) lines due to a somatic mutation of the phosphatidylinositol glycan anchor (PIG-A) gene located on Xp22.1. PNH incidence is 1.5-2 cases per million of the population per year. PNH can affect multiple systems in the body and requires multidisciplinary clinical management. Patients can manifest with severe pancytopenia, life-threatening thrombosis affecting the hepatic, abdominal, cerebral, and subdermal veins, and high requirements for blood transfusion due to haemolytic anemia. PNH can also be associated with bone marrow failure. Advances in diagnostic techniques and a targeted therapeutic approach for PNH have emerged in the last two decades. Eculizumab, a promising humanized monoclonal antibody against C5, is the first approved therapy for PNH.
Keywords: Paroxysmal nocturnal hemoglobinuria; diagnosis; eculizumab; treatment.
Figures


References
-
- Inoue N, Izui-Sarumaru T, Murakami Y, Endo Y, Nishimura J, Kurokawa K, Kuwayama M, Shime H, Machii T, Kanakura Y, Meyers G, Wittwer C, Chen Z, Babcock W, Frei-Lahr D, Parker CJ, Kinoshita T. Molecular basis of clonal expansion of hematopoiesis in 2 patients with paroxysmal nocturnal hemoglobinuria (PNH) Blood. 2006;108:4232–4236. - PMC - PubMed
-
- Takeda J, Miyata T, Kawagoe K, Iida Y, Endo Y, Fujita T, Takahashi M, Kitani T, Kinoshita T. Deficiency of the GPI anchor caused by a somatic mutation of the PIG-A gene in paroxysmal nocturnal hemoglobinuria. Cell. 1993;73:703–711. - PubMed
-
- de Latour RP, Mary JY, Salanoubat C, Terriou L, Etienne G, Mohty M, Roth S, de Guibert S, Maury S, Cahn JY, Socié G French Society of Hematology; French Association of Young Hematologists. Paroxysmal nocturnal hemoglobinuria: natural history of disease subcategories. Blood. 2008;112:3099–3106. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous
