Moles of a Substance per Cell Is a Highly Informative Dosing Metric in Cell Culture
- PMID: 26172833
- PMCID: PMC4501792
- DOI: 10.1371/journal.pone.0132572
Moles of a Substance per Cell Is a Highly Informative Dosing Metric in Cell Culture
Abstract
Background: The biological consequences upon exposure of cells in culture to a dose of xenobiotic are not only dependent on biological variables, but also the physical aspects of experiments e.g. cell number and media volume. Dependence on physical aspects is often overlooked due to the unrecognized ambiguity in the dominant metric used to express exposure, i.e. initial concentration of xenobiotic delivered to the culture medium over the cells. We hypothesize that for many xenobiotics, specifying dose as moles per cell will reduce this ambiguity. Dose as moles per cell can also provide additional information not easily obtainable with traditional dosing metrics.
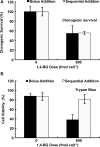
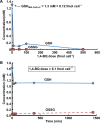
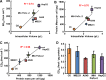
Methods: Here, 1,4-benzoquinone and oligomycin A are used as model compounds to investigate moles per cell as an informative dosing metric. Mechanistic insight into reactions with intracellular molecules, differences between sequential and bolus addition of xenobiotic and the influence of cell volume and protein content on toxicity are also investigated.
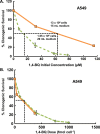
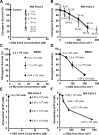
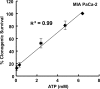
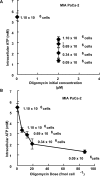
Results: When the dose of 1,4-benzoquinone or oligomycin A was specified as moles per cell, toxicity was independent of the physical conditions used (number of cells, volume of medium). When using moles per cell as a dose-metric, direct quantitative comparisons can be made between biochemical or biological endpoints and the dose of xenobiotic applied. For example, the toxicity of 1,4-benzoquinone correlated inversely with intracellular volume for all five cell lines exposed (C6, MDA-MB231, A549, MIA PaCa-2, and HepG2).
Conclusions: Moles per cell is a useful and informative dosing metric in cell culture. This dosing metric is a scalable parameter that: can reduce ambiguity between experiments having different physical conditions; provides additional mechanistic information; allows direct comparison between different cells; affords a more uniform platform for experimental design; addresses the important issue of repeatability of experimental results, and could increase the translatability of information gained from in vitro experiments.
Conflict of interest statement
Figures
Similar articles
-
Measuring and modeling of binary mixture effects of pharmaceuticals and nickel on cell viability/cytotoxicity in the human hepatoma derived cell line HepG2.Toxicol Appl Pharmacol. 2010 May 1;244(3):336-43. doi: 10.1016/j.taap.2010.01.012. Epub 2010 Feb 2. Toxicol Appl Pharmacol. 2010. PMID: 20132835
-
Biomarkers in aquatic plants: selection and utility.Rev Environ Contam Toxicol. 2009;198:49-109. doi: 10.1007/978-0-387-09647-6_2. Rev Environ Contam Toxicol. 2009. PMID: 19253039 Review.
-
Evaluation of the metabolism and hepatotoxicity of xenobiotics utilizing precision-cut slices.Xenobiotica. 2013 Jan;43(1):41-53. doi: 10.3109/00498254.2012.734643. Epub 2012 Nov 6. Xenobiotica. 2013. PMID: 23131042 Review.
-
Toxicity profiles of four metals and 17 xenobiotics in the human hepatoma cell line HepG2 and the protozoa Tetrahymena pyriformis--a comparison.Environ Toxicol. 2011 Apr;26(2):171-86. doi: 10.1002/tox.20541. Environ Toxicol. 2011. PMID: 19790250
-
The delivered dose: Applying particokinetics to in vitro investigations of nanoparticle internalization by macrophages.J Control Release. 2012 Sep 10;162(2):259-66. doi: 10.1016/j.jconrel.2012.07.019. Epub 2012 Jul 21. J Control Release. 2012. PMID: 22824784
Cited by
-
Effective Drug Concentration and Selectivity Depends on Fraction of Primitive Cells.Int J Mol Sci. 2021 May 6;22(9):4931. doi: 10.3390/ijms22094931. Int J Mol Sci. 2021. PMID: 34066491 Free PMC article.
-
The Xc- inhibitor sulfasalazine improves the anti-cancer effect of pharmacological vitamin C in prostate cancer cells via a glutathione-dependent mechanism.Cell Oncol (Dordr). 2020 Feb;43(1):95-106. doi: 10.1007/s13402-019-00474-8. Epub 2019 Oct 15. Cell Oncol (Dordr). 2020. PMID: 31617161
-
Superoxide Dismutase Mimetic GC4419 Enhances the Oxidation of Pharmacological Ascorbate and Its Anticancer Effects in an H₂O₂-Dependent Manner.Antioxidants (Basel). 2018 Jan 19;7(1):18. doi: 10.3390/antiox7010018. Antioxidants (Basel). 2018. PMID: 29351198 Free PMC article.
-
Quantitation of spin probe-detectable oxidants in cells using electron paramagnetic resonance spectroscopy: To probe or to trap?Free Radic Biol Med. 2020 Jul;154:84-94. doi: 10.1016/j.freeradbiomed.2020.04.020. Epub 2020 May 4. Free Radic Biol Med. 2020. PMID: 32376456 Free PMC article.
-
O2⋅- and H2O2-Mediated Disruption of Fe Metabolism Causes the Differential Susceptibility of NSCLC and GBM Cancer Cells to Pharmacological Ascorbate.Cancer Cell. 2017 Apr 10;31(4):487-500.e8. doi: 10.1016/j.ccell.2017.02.018. Epub 2017 Mar 30. Cancer Cell. 2017. PMID: 28366679 Free PMC article. Clinical Trial.
References
-
- Russel WMS, Burch RL. The principles of humane experimental technique. London: Methuen; 1959. Full text available at: http://altweb.jhsph.edu/pubs/books/humane_exp/het-toc accessed 2015.06.01
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

