Pharmacological inhibition of apical sodium-dependent bile acid transporter changes bile composition and blocks progression of sclerosing cholangitis in multidrug resistance 2 knockout mice
- PMID: 26172874
- PMCID: PMC4713368
- DOI: 10.1002/hep.27973
Pharmacological inhibition of apical sodium-dependent bile acid transporter changes bile composition and blocks progression of sclerosing cholangitis in multidrug resistance 2 knockout mice
Abstract
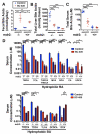
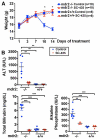
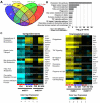
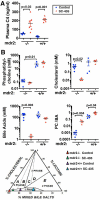
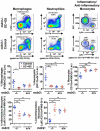
Deficiency of multidrug resistance 2 (mdr2), a canalicular phospholipid floppase, leads to excretion of low-phospholipid "toxic" bile causing progressive cholestasis. We hypothesize that pharmacological inhibition of the ileal, apical sodium-dependent bile acid transporter (ASBT), blocks progression of sclerosing cholangitis in mdr2(-/-) mice. Thirty-day-old, female mdr2(-/-) mice were fed high-fat chow containing 0.006% SC-435, a minimally absorbed, potent inhibitor of ASBT, providing, on average, 11 mg/kg/day of compound. Bile acids (BAs) and phospholipids were measured by mass spectrometry. Compared with untreated mdr2(-/-) mice, SC-435 treatment for 14 days increased fecal BA excretion by 8-fold, lowered total BA concentration in liver by 65%, reduced total BA and individual hydrophobic BA concentrations in serum by >98%, and decreased plasma alanine aminotransferase, total bilirubin, and serum alkaline phosphatase levels by 86%, 93%, and 55%, respectively. Liver histology of sclerosing cholangitis improved, and extent of fibrosis decreased concomitant with reduction of hepatic profibrogenic gene expression. Biliary BA concentrations significantly decreased and phospholipids remained low and unchanged with treatment. The phosphatidylcholine (PC)/BA ratio in treated mice corrected toward a ratio of 0.28 found in wild-type mice, indicating decreased bile toxicity. Hepatic RNA sequencing studies revealed up-regulation of putative anti-inflammatory and antifibrogenic genes, including Ppara and Igf1, and down-regulation of several proinflammatory genes, including Ccl2 and Lcn2, implicated in leukocyte recruitment. Flow cytometric analysis revealed significant reduction of frequencies of hepatic CD11b(+) F4/80(+) Kupffer cells and CD11b(+) Gr1(+) neutrophils, accompanied by expansion of anti-inflammatory Ly6C(-) monocytes in treated mdr2(-/-) mice.
Conclusion: Inhibition of ASBT reduces BA pool size and retention of hydrophobic BA, favorably alters the biliary PC/BA ratio, profoundly changes the hepatic transcriptome, attenuates recruitment of leukocytes, and abrogates progression of murine sclerosing cholangitis.
© 2015 by the American Association for the Study of Liver Diseases.
Figures


Comment in
-
Toxic bile and sclerosing cholangitis: Is there a role for pharmacological interruption of the bile acid enterohepatic circulation?Hepatology. 2016 Feb;63(2):363-4. doi: 10.1002/hep.28363. Epub 2016 Jan 6. Hepatology. 2016. PMID: 26600416 Free PMC article. No abstract available.
Similar articles
-
Inhibition of intestinal bile acid absorption improves cholestatic liver and bile duct injury in a mouse model of sclerosing cholangitis.J Hepatol. 2016 Mar;64(3):674-81. doi: 10.1016/j.jhep.2015.10.024. Epub 2015 Oct 31. J Hepatol. 2016. PMID: 26529078
-
Toxic bile and sclerosing cholangitis: Is there a role for pharmacological interruption of the bile acid enterohepatic circulation?Hepatology. 2016 Feb;63(2):363-4. doi: 10.1002/hep.28363. Epub 2016 Jan 6. Hepatology. 2016. PMID: 26600416 Free PMC article. No abstract available.
-
Loss of apical sodium bile acid transporter alters bile acid circulation and reduces biliary damage in cholangitis.Am J Physiol Gastrointest Liver Physiol. 2023 Jan 1;324(1):G60-G77. doi: 10.1152/ajpgi.00112.2022. Epub 2022 Nov 21. Am J Physiol Gastrointest Liver Physiol. 2023. PMID: 36410025 Free PMC article.
-
Lessons from the toxic bile concept for the pathogenesis and treatment of cholestatic liver diseases.Wien Med Wochenschr. 2008;158(19-20):542-8. doi: 10.1007/s10354-008-0592-1. Wien Med Wochenschr. 2008. PMID: 18998069 Review.
-
Apical Sodium-Dependent Transporter Inhibitors in Primary Biliary Cholangitis and Primary Sclerosing Cholangitis.Dig Dis. 2017;35(3):267-274. doi: 10.1159/000450988. Epub 2017 Mar 1. Dig Dis. 2017. PMID: 28249258 Review.
Cited by
-
Role of bile acids and their receptors in gastrointestinal and hepatic pathophysiology.Nat Rev Gastroenterol Hepatol. 2022 Jul;19(7):432-450. doi: 10.1038/s41575-021-00566-7. Epub 2022 Feb 14. Nat Rev Gastroenterol Hepatol. 2022. PMID: 35165436 Review.
-
Interim results from an ongoing, open-label, single-arm trial of odevixibat in progressive familial intrahepatic cholestasis.JHEP Rep. 2023 Apr 29;5(8):100782. doi: 10.1016/j.jhepr.2023.100782. eCollection 2023 Aug. JHEP Rep. 2023. PMID: 37456676 Free PMC article.
-
Current and future therapies for inherited cholestatic liver diseases.World J Gastroenterol. 2017 Feb 7;23(5):763-775. doi: 10.3748/wjg.v23.i5.763. World J Gastroenterol. 2017. PMID: 28223721 Free PMC article. Review.
-
Therapeutic potential of berberine in attenuating cholestatic liver injury: insights from a PSC mouse model.Cell Biosci. 2024 Jan 25;14(1):14. doi: 10.1186/s13578-024-01195-8. Cell Biosci. 2024. PMID: 38273376 Free PMC article.
-
Emerging therapies for PBC.J Gastroenterol. 2020 Mar;55(3):261-272. doi: 10.1007/s00535-020-01664-0. Epub 2020 Jan 22. J Gastroenterol. 2020. PMID: 31970467 Free PMC article. Review.
References
-
- Beuers U, Trauner M, Jansen P, Poupon R. New paradigms in the treatment of hepatic cholestasis: From UDCA to FXR, PXR and beyond. J Hepatol. 2015;62:S25–S37. - PubMed
-
- Hirschfield GM, Heathcote EJ, Gershwin ME. Pathogenesis of cholestatic liver disease and therapeutic approaches. Gastroenterology. 2010;139:1481–1496. - PubMed
-
- Patel T, Gores GJ. Apoptosis and hepatobiliary disease. Hepatology. 1995;21:1725–1741. - PubMed
-
- Karlsen TH, Franke A, Melum E, Kaser A, Hov JR, Balschun T, Lie BA, et al. Genome-wide association analysis in primary sclerosing cholangitis. Gastroenterology. 2010;138:1102–1111. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

