The amyloid precursor protein (APP) intracellular domain regulates translation of p44, a short isoform of p53, through an IRES-dependent mechanism
- PMID: 26174856
- PMCID: PMC4562799
- DOI: 10.1016/j.neurobiolaging.2015.06.021
The amyloid precursor protein (APP) intracellular domain regulates translation of p44, a short isoform of p53, through an IRES-dependent mechanism
Abstract
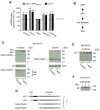
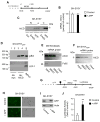
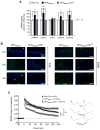
p44 is a short isoform of the tumor suppressor protein p53 that is regulated in an age-dependent manner. When overexpressed in the mouse, it causes a progeroid phenotype that includes premature cognitive decline, synaptic defects, and hyperphosphorylation of tau. The hyperphosphorylation of tau has recently been linked to the ability of p44 to regulate transcription of relevant tau kinases. Here, we report that the amyloid precursor protein (APP) intracellular domain (AICD), which results from the processing of the APP, regulates translation of p44 through a cap-independent mechanism that requires direct binding to the second internal ribosome entry site (IRES) of the p53 mRNA. We also report that AICD associates with nucleolin, an already known IRES-specific trans-acting factor that binds with p53 IRES elements and regulates translation of p53 isoforms. The potential biological impact of our findings was assessed in a mouse model of Alzheimer's disease. In conclusion, our study reveals a novel aspect of AICD and p53/p44 biology and provides a possible molecular link between APP, p44, and tau.
Keywords: AICD; Aging; Alzheimer's disease; IRES; p44; p53.
Copyright © 2015 Elsevier Inc. All rights reserved.
Conflict of interest statement
None of the authors report any potential or actual conflict of interest.
Figures


Similar articles
-
P44, the 'longevity-assurance' isoform of P53, regulates tau phosphorylation and is activated in an age-dependent fashion.Aging Cell. 2014 Jun;13(3):449-56. doi: 10.1111/acel.12192. Epub 2014 Feb 25. Aging Cell. 2014. PMID: 24341977 Free PMC article.
-
Effects of curcumin and demethoxycurcumin on amyloid-β precursor and tau proteins through the internal ribosome entry sites: a potential therapeutic for Alzheimer's disease.Taiwan J Obstet Gynecol. 2012 Dec;51(4):554-64. doi: 10.1016/j.tjog.2012.09.010. Taiwan J Obstet Gynecol. 2012. PMID: 23276558
-
Altered longevity-assurance activity of p53:p44 in the mouse causes memory loss, neurodegeneration and premature death.Aging Cell. 2010 Apr;9(2):174-90. doi: 10.1111/j.1474-9726.2010.00547.x. Aging Cell. 2010. PMID: 20409077 Free PMC article.
-
Modeling Alzheimer's disease in transgenic mice: effect of age and of presenilin1 on amyloid biochemistry and pathology in APP/London mice.Exp Gerontol. 2000 Sep;35(6-7):831-41. doi: 10.1016/s0531-5565(00)00149-2. Exp Gerontol. 2000. PMID: 11053674 Review.
-
Targeting IRES-Mediated p53 Synthesis for Cancer Diagnosis and Therapeutics.Int J Mol Sci. 2017 Jan 4;18(1):93. doi: 10.3390/ijms18010093. Int J Mol Sci. 2017. PMID: 28054974 Free PMC article. Review.
Cited by
-
Alternative Mechanisms of p53 Action During the Unfolded Protein Response.Cancers (Basel). 2020 Feb 10;12(2):401. doi: 10.3390/cancers12020401. Cancers (Basel). 2020. PMID: 32050651 Free PMC article. Review.
-
The AICD interactome: implications in neurodevelopment and neurodegeneration.Biochem Soc Trans. 2024 Dec 19;52(6):2539-2556. doi: 10.1042/BST20241510. Biochem Soc Trans. 2024. PMID: 39670668 Free PMC article. Review.
-
The β-Secretase 1 Enzyme as a Novel Therapeutic Target for Prostate Cancer.Cancers (Basel). 2023 Dec 19;16(1):10. doi: 10.3390/cancers16010010. Cancers (Basel). 2023. PMID: 38201438 Free PMC article.
-
The pleiotropic role of p53 in functional/dysfunctional neurons: focus on pathogenesis and diagnosis of Alzheimer's disease.Alzheimers Res Ther. 2020 Dec 3;12(1):160. doi: 10.1186/s13195-020-00732-0. Alzheimers Res Ther. 2020. PMID: 33272326 Free PMC article. Review.
-
Internal Ribosome Entry Site (IRES)-Mediated Translation and Its Potential for Novel mRNA-Based Therapy Development.Biomedicines. 2022 Aug 2;10(8):1865. doi: 10.3390/biomedicines10081865. Biomedicines. 2022. PMID: 36009412 Free PMC article. Review.
References
-
- Beckett C, Nalivaeva NN, Belyaev ND, Turner AJ. Nuclear signalling by membrane protein intracellular domains: the AICD enigma. Cellular signalling. 2012;24(2):402–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

