Role of RAGE in Alzheimer's Disease
- PMID: 26175217
- PMCID: PMC11482350
- DOI: 10.1007/s10571-015-0233-3
Role of RAGE in Alzheimer's Disease
Abstract
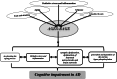
Receptor for advanced glycation end products (RAGE) is a receptor of the immunoglobulin super family that plays various important roles under physiological and pathological conditions. Compelling evidence suggests that RAGE acts as both an inflammatory intermediary and a critical inducer of oxidative stress, underlying RAGE-induced Alzheimer-like pathophysiological changes that drive the process of Alzheimer's disease (AD). A critical role of RAGE in AD includes beta-amyloid (Aβ) production and accumulation, the formation of neurofibrillary tangles, failure of synaptic transmission, and neuronal degeneration. The steady-state level of Aβ depends on the balance between production and clearance. RAGE plays an important role in the Aβ clearance. RAGE acts as an important transporter via regulating influx of circulating Aβ into brain, whereas the efflux of brain-derived Aβ into the circulation via BBB is implemented by LRP1. RAGE could be an important contributor to Aβ generation via enhancing the activity of β- and/or γ-secretases and activating inflammatory response and oxidative stress. However, sRAGE-Aβ interactions could inhibit Aβ neurotoxicity and promote Aβ clearance from brain. Meanwhile, RAGE could be a promoting factor for the synaptic dysfunction and neuronal circuit dysfunction which are both the material structure of cognition, and the physiological and pathological basis of cognition. In addition, RAGE could be a trigger for the pathogenesis of Aβ and tau hyper-phosphorylation which both participate in the process of cognitive impairment. Preclinical and clinical studies have supported that RAGE inhibitors could be useful in the treatment of AD. Thus, an effective measure to inhibit RAGE may be a novel drug target in AD.
Keywords: Alzheimer’s disease; Beta-amyloid; Cognitive impairment; Receptor for advanced glycation end products; Tau hyperphosphorylation.
Conflict of interest statement
None declared.
Figures

Similar articles
-
AGE-RAGE stress: a changing landscape in pathology and treatment of Alzheimer's disease.Mol Cell Biochem. 2019 Sep;459(1-2):95-112. doi: 10.1007/s11010-019-03553-4. Epub 2019 May 11. Mol Cell Biochem. 2019. PMID: 31079281 Review.
-
Receptor for Advanced Glycation End Products: Dementia and Cognitive Impairment.Drug Res (Stuttg). 2023 Jun;73(5):247-250. doi: 10.1055/a-2015-8041. Epub 2023 Mar 8. Drug Res (Stuttg). 2023. PMID: 36889338 Review.
-
Roles of the Receptor for Advanced Glycation End Products and Its Ligands in the Pathogenesis of Alzheimer's Disease.Int J Mol Sci. 2025 Jan 5;26(1):403. doi: 10.3390/ijms26010403. Int J Mol Sci. 2025. PMID: 39796257 Free PMC article. Review.
-
Receptor for advanced glycation end products mediates sepsis-triggered amyloid-β accumulation, Tau phosphorylation, and cognitive impairment.J Biol Chem. 2018 Jan 5;293(1):226-244. doi: 10.1074/jbc.M117.786756. Epub 2017 Nov 10. J Biol Chem. 2018. PMID: 29127203 Free PMC article.
-
Effect of High Cholesterol Regulation of LRP1 and RAGE on Aβ Transport Across the Blood-Brain Barrier in Alzheimer's Disease.Curr Alzheimer Res. 2021;18(5):428-442. doi: 10.2174/1567205018666210906092940. Curr Alzheimer Res. 2021. PMID: 34488598
Cited by
-
Tanshinone IIA attenuates neuroinflammation via inhibiting RAGE/NF-κB signaling pathway in vivo and in vitro.J Neuroinflammation. 2020 Oct 14;17(1):302. doi: 10.1186/s12974-020-01981-4. J Neuroinflammation. 2020. PMID: 33054814 Free PMC article.
-
Overexpression of the receptor for advanced glycation end-products in the auditory cortex of rats with noise-induced hearing loss.BMC Neurosci. 2021 May 21;22(1):38. doi: 10.1186/s12868-021-00642-3. BMC Neurosci. 2021. PMID: 34020590 Free PMC article.
-
Chronic Sleep Restriction Induces Aβ Accumulation by Disrupting the Balance of Aβ Production and Clearance in Rats.Neurochem Res. 2019 Apr;44(4):859-873. doi: 10.1007/s11064-019-02719-2. Epub 2019 Jan 10. Neurochem Res. 2019. PMID: 30632087
-
Compromised DNA repair is responsible for diabetes-associated fibrosis.EMBO J. 2020 Jun 2;39(11):e103477. doi: 10.15252/embj.2019103477. Epub 2020 Apr 27. EMBO J. 2020. PMID: 32338774 Free PMC article.
-
Inflammation in CNS neurodegenerative diseases.Immunology. 2018 Jun;154(2):204-219. doi: 10.1111/imm.12922. Epub 2018 Apr 17. Immunology. 2018. PMID: 29513402 Free PMC article. Review.
References
-
- Alves J, Magalhaes R, Arantes M, Cruz S, Goncalves OF, Sampaio A (2015) Cognitive rehabilitation in a visual variant of Alzheimer’s disease. Appl Neuropsychol Adult 22:73–78 - PubMed
-
- Baiguera S, Fioravanzo L, Grandi C, Di LR, Parnigotto PP, Folin M (2009) Involvement of the receptor for advanced glycation-end products (RAGE) in beta-amyloid-induced toxic effects in rat cerebromicrovascular endothelial cells cultured in vitro. Int J Mol Med 24:9–15 - PubMed
-
- Barroso E, Del Valle J, Porquet D, Vieira Santos AM, Salvado L, Rodriguez-Rodriguez R, Gutierrez P, Anglada-Huguet M, Alberch J, Camins A et al (2013) Tau hyperphosphorylation and increased BACE1 and RAGE levels in the cortex of PPARbeta/delta-null mice. Biochim Biophys Acta 1832:1241–1248 - PubMed
-
- Bellucci A, Rosi MC, Grossi C, Fiorentini A, Luccarini I, Casamenti F (2007) Abnormal processing of tau in the brain of aged TgCRND8 mice. Neurobiol Dis 27:328–338 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

