Role of gut microbiota in the modulation of atherosclerosis-associated immune response
- PMID: 26175728
- PMCID: PMC4485310
- DOI: 10.3389/fmicb.2015.00671
Role of gut microbiota in the modulation of atherosclerosis-associated immune response
Abstract
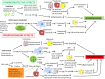
Inflammation and metabolic abnormalities are linked to each other. At present, pathogenic inflammatory response was recognized as a major player in metabolic diseases. In humans, intestinal microflora could significantly influence the development of metabolic diseases including atherosclerosis. Commensal bacteria were shown to activate inflammatory pathways through altering lipid metabolism in adipocytes, macrophages, and vascular cells, inducing insulin resistance, and producing trimethylamine-N-oxide. However, gut microbiota could also play the atheroprotective role associated with anthocyanin metabolism and administration of probiotics and their components. Here, we review the mechanisms by which the gut microbiota may influence atherogenesis.
Keywords: arteries; atherogenesis; atherosclerosis; immune system; intestinal microbiota.
Figures
References
-
- Amar J., Burcelin R., Ruidavets J. B., Cani P. D., Fauvel J., Alessi M. C., et al. (2008). Energy intake is associated with endotoxemia in apparently healthy men. Am. J. Clin. Nutr. 87 1219–1223. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

