Stem cell therapy and tissue engineering for correction of congenital heart disease
- PMID: 26176009
- PMCID: PMC4485350
- DOI: 10.3389/fcell.2015.00039
Stem cell therapy and tissue engineering for correction of congenital heart disease
Abstract
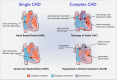
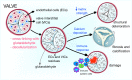
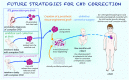
This review article reports on the new field of stem cell therapy and tissue engineering and its potential on the management of congenital heart disease. To date, stem cell therapy has mainly focused on treatment of ischemic heart disease and heart failure, with initial indication of safety and mild-to-moderate efficacy. Preclinical studies and initial clinical trials suggest that the approach could be uniquely suited for the correction of congenital defects of the heart. The basic concept is to create living material made by cellularized grafts that, once implanted into the heart, grows and remodels in parallel with the recipient organ. This would make a substantial improvement in current clinical management, which often requires repeated surgical corrections for failure of implanted grafts. Different types of stem cells have been considered and the identification of specific cardiac stem cells within the heterogeneous population of mesenchymal and stromal cells offers opportunities for de novo cardiomyogenesis. In addition, endothelial cells and vascular progenitors, including cells with pericyte characteristics, may be necessary to generate efficiently perfused grafts. The implementation of current surgical grafts by stem cell engineering could address the unmet clinical needs of patients with congenital heart defects.
Keywords: biomaterial; congenital heart disease; scaffold; stem cells; tissue engineering.
Figures


Similar articles
-
Potential for stem cell use in congenital heart disease.Future Cardiol. 2012 Mar;8(2):161-9. doi: 10.2217/fca.12.13. Future Cardiol. 2012. PMID: 22413976 Review.
-
Thymus-Derived Mesenchymal Stem Cells for Tissue Engineering Clinical-Grade Cardiovascular Grafts.Tissue Eng Part A. 2018 May;24(9-10):794-808. doi: 10.1089/ten.TEA.2017.0290. Epub 2017 Nov 30. Tissue Eng Part A. 2018. PMID: 29054134
-
Expansion and characterization of neonatal cardiac pericytes provides a novel cellular option for tissue engineering in congenital heart disease.J Am Heart Assoc. 2015 Jun 16;4(6):e002043. doi: 10.1161/JAHA.115.002043. J Am Heart Assoc. 2015. PMID: 26080813 Free PMC article.
-
Wharton's Jelly-Mesenchymal Stem Cell-Engineered Conduit for Pediatric Translation in Heart Defect.Tissue Eng Part A. 2021 Feb;27(3-4):201-213. doi: 10.1089/ten.TEA.2020.0088. Epub 2020 Jul 31. Tissue Eng Part A. 2021. PMID: 32571164
-
Organ reconstruction: Dream or reality for the future.Biomed Mater Eng. 2017;28(s1):S121-S127. doi: 10.3233/BME-171633. Biomed Mater Eng. 2017. PMID: 28372287 Review.
Cited by
-
Natural Polymers in Heart Valve Tissue Engineering: Strategies, Advances and Challenges.Biomedicines. 2022 May 8;10(5):1095. doi: 10.3390/biomedicines10051095. Biomedicines. 2022. PMID: 35625830 Free PMC article. Review.
-
The Potential Impact and Timeline of Engineering on Congenital Interventions.Pediatr Cardiol. 2020 Mar;41(3):522-538. doi: 10.1007/s00246-020-02335-w. Epub 2020 Mar 20. Pediatr Cardiol. 2020. PMID: 32198587 Review.
-
Stem cell therapy targeting the right ventricle in pulmonary arterial hypertension: is it a potential avenue of therapy?Pulm Circ. 2018 Apr-Jun;8(2):2045893218755979. doi: 10.1177/2045893218755979. Epub 2018 Feb 26. Pulm Circ. 2018. PMID: 29480154 Free PMC article.
-
Direct Reprogramming of Cardiac Fibroblasts to Repair the Injured Heart.J Cardiovasc Dev Dis. 2021 Jun 22;8(7):72. doi: 10.3390/jcdd8070072. J Cardiovasc Dev Dis. 2021. PMID: 34206355 Free PMC article. Review.
-
Cell Sources for Tissue Engineering Strategies to Treat Calcific Valve Disease.Front Cardiovasc Med. 2018 Nov 6;5:155. doi: 10.3389/fcvm.2018.00155. eCollection 2018. Front Cardiovasc Med. 2018. PMID: 30460245 Free PMC article. Review.
References
-
- Avolio E., Meloni M., Spencer H. L., Riu F., Katare R., Mangialardi G., et al. . (2015a). Combined intramyocardial delivery of human pericytes and cardiac stem cells additively improves the healing of mouse infarcted hearts through stimulation of vascular and muscular repair. Circ. Res. 116, e81–e94. 10.1161/CIRCRESAHA.115.306146 - DOI - PubMed
-
- Avolio E., Rodriguez-Arabaolaza I., Spencer H., Riu F., Mangialardi G., Slater S., et al. . (2015b). Expansion and characterization of neonatal cardiac pericytes provides a novel cellular option for tissue engineering in congenital heart disease. J. Am. Heart Assoc. 4:e002043. 10.1161/JAHA.115.002043 - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

