Development of the endocrine pancreas and novel strategies for β-cell mass restoration and diabetes therapy
- PMID: 26176316
- PMCID: PMC4568803
- DOI: 10.1590/1414-431X20154363
Development of the endocrine pancreas and novel strategies for β-cell mass restoration and diabetes therapy
Abstract
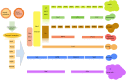
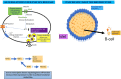
Diabetes mellitus represents a serious public health problem owing to its global prevalence in the last decade. The causes of this metabolic disease include dysfunction and/or insufficient number of β cells. Existing diabetes mellitus treatments do not reverse or control the disease. Therefore, β-cell mass restoration might be a promising treatment. Several restoration approaches have been developed: inducing the proliferation of remaining insulin-producing cells, de novo islet formation from pancreatic progenitor cells (neogenesis), and converting non-β cells within the pancreas to β cells (transdifferentiation) are the most direct, simple, and least invasive ways to increase β-cell mass. However, their clinical significance is yet to be determined. Hypothetically, β cells or islet transplantation methods might be curative strategies for diabetes mellitus; however, the scarcity of donors limits the clinical application of these approaches. Thus, alternative cell sources for β-cell replacement could include embryonic stem cells, induced pluripotent stem cells, and mesenchymal stem cells. However, most differentiated cells obtained using these techniques are functionally immature and show poor glucose-stimulated insulin secretion compared with native β cells. Currently, their clinical use is still hampered by ethical issues and the risk of tumor development post transplantation. In this review, we briefly summarize the current knowledge of mouse pancreas organogenesis, morphogenesis, and maturation, including the molecular mechanisms involved. We then discuss two possible approaches of β-cell mass restoration for diabetes mellitus therapy: β-cell regeneration and β-cell replacement. We critically analyze each strategy with respect to the accessibility of the cells, potential risk to patients, and possible clinical outcomes.
Figures

Similar articles
-
Concise review: pancreas regeneration: recent advances and perspectives.Stem Cells Transl Med. 2012 Feb;1(2):150-9. doi: 10.5966/sctm.2011-0025. Epub 2012 Jan 26. Stem Cells Transl Med. 2012. PMID: 23197762 Free PMC article. Review.
-
How to make insulin-producing pancreatic β cells for diabetes treatment.Sci China Life Sci. 2017 Mar;60(3):239-248. doi: 10.1007/s11427-016-0211-3. Epub 2016 Oct 27. Sci China Life Sci. 2017. PMID: 27796637 Review.
-
New sources of beta-cells for treating diabetes.J Endocrinol. 2009 Jul;202(1):13-6. doi: 10.1677/JOE-09-0097. Epub 2009 May 6. J Endocrinol. 2009. PMID: 19420008 Review.
-
Insulin-producing cells derived from stem cells: recent progress and future directions.J Cell Mol Med. 2006 Oct-Dec;10(4):866-83. doi: 10.1111/j.1582-4934.2006.tb00531.x. J Cell Mol Med. 2006. PMID: 17125591 Review.
-
[Cellular therapy of diabetes: focus on the latest developments].Med Sci (Paris). 2016 Apr;32(4):401-7. doi: 10.1051/medsci/20163204019. Epub 2016 May 2. Med Sci (Paris). 2016. PMID: 27137698 Review. French.
Cited by
-
Valproic Acid Exposure of Pregnant Rats During Organogenesis Disturbs Pancreas Development in Insulin Synthesis and Secretion of the Offspring.Toxicol Res. 2018 Apr;34(2):173-182. doi: 10.5487/TR.2018.34.2.173. Epub 2018 Apr 15. Toxicol Res. 2018. PMID: 29686779 Free PMC article.
-
In vitro generation of transplantable insulin-producing cells from canine adipose-derived mesenchymal stem cells.Sci Rep. 2022 Jun 1;12(1):9127. doi: 10.1038/s41598-022-13114-3. Sci Rep. 2022. PMID: 35650303 Free PMC article.
-
FTIR Spectroscopic and Molecular Analysis during Differentiation of Pluripotent Stem Cells to Pancreatic Cells.Stem Cells Int. 2016;2016:6709714. doi: 10.1155/2016/6709714. Epub 2016 Aug 29. Stem Cells Int. 2016. PMID: 27651798 Free PMC article.
-
Development and Characteristics of Pancreatic Epsilon Cells.Int J Mol Sci. 2019 Apr 16;20(8):1867. doi: 10.3390/ijms20081867. Int J Mol Sci. 2019. PMID: 31014006 Free PMC article. Review.
-
Efficiency of Bone Marrow-Derived Mesenchymal Stem Cells and Hesperetin in the Treatment of Streptozotocin-Induced Type 1 Diabetes in Wistar Rats.Pharmaceuticals (Basel). 2023 Jun 8;16(6):859. doi: 10.3390/ph16060859. Pharmaceuticals (Basel). 2023. PMID: 37375806 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

