Identification of I-7 expands the repertoire of genes for resistance to Fusarium wilt in tomato to three resistance gene classes
- PMID: 26177154
- PMCID: PMC6638478
- DOI: 10.1111/mpp.12294
Identification of I-7 expands the repertoire of genes for resistance to Fusarium wilt in tomato to three resistance gene classes
Abstract
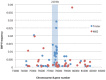
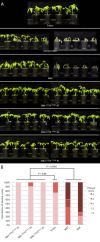
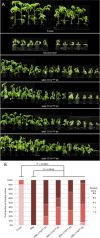
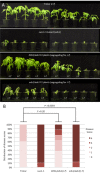
The tomato I-3 and I-7 genes confer resistance to Fusarium oxysporum f. sp. lycopersici (Fol) race 3 and were introgressed into the cultivated tomato, Solanum lycopersicum, from the wild relative Solanum pennellii. I-3 has been identified previously on chromosome 7 and encodes an S-receptor-like kinase, but little is known about I-7. Molecular markers have been developed for the marker-assisted breeding of I-3, but none are available for I-7. We used an RNA-seq and single nucleotide polymorphism (SNP) analysis approach to map I-7 to a small introgression of S. pennellii DNA (c. 210 kb) on chromosome 8, and identified I-7 as a gene encoding a leucine-rich repeat receptor-like protein (LRR-RLP), thereby expanding the repertoire of resistance protein classes conferring resistance to Fol. Using an eds1 mutant of tomato, we showed that I-7, like many other LRR-RLPs conferring pathogen resistance in tomato, is EDS1 (Enhanced Disease Susceptibility 1) dependent. Using transgenic tomato plants carrying only the I-7 gene for Fol resistance, we found that I-7 also confers resistance to Fol races 1 and 2. Given that Fol race 1 carries Avr1, resistance to Fol race 1 indicates that I-7-mediated resistance, unlike I-2- or I-3-mediated resistance, is not suppressed by Avr1. This suggests that Avr1 is not a general suppressor of Fol resistance in tomato, leading us to hypothesize that Avr1 may be acting against an EDS1-independent pathway for resistance activation. The identification of I-7 has allowed us to develop molecular markers for marker-assisted breeding of both genes currently known to confer Fol race 3 resistance (I-3 and I-7). Given that I-7-mediated resistance is not suppressed by Avr1, I-7 may be a useful addition to I-3 in the tomato breeder's toolbox.
Keywords: Fusarium oxysporum f. sp. lycopersici; Fusarium wilt; Solanum lycopersicum; Solanum pennellii; leucine-rich repeat; plant disease resistance gene; receptor-like protein.
© 2015 BSPP and John Wiley & Sons Ltd.
Figures


Similar articles
-
The tomato I-3 gene: a novel gene for resistance to Fusarium wilt disease.New Phytol. 2015 Jul;207(1):106-118. doi: 10.1111/nph.13348. Epub 2015 Mar 4. New Phytol. 2015. PMID: 25740416
-
The tomato I gene for Fusarium wilt resistance encodes an atypical leucine-rich repeat receptor-like protein whose function is nevertheless dependent on SOBIR1 and SERK3/BAK1.Plant J. 2017 Mar;89(6):1195-1209. doi: 10.1111/tpj.13458. Epub 2017 Feb 11. Plant J. 2017. PMID: 27995670
-
Multiple Evolutionary Trajectories Have Led to the Emergence of Races in Fusarium oxysporum f. sp. lycopersici.Appl Environ Microbiol. 2017 Feb 1;83(4):e02548-16. doi: 10.1128/AEM.02548-16. Print 2017 Feb 15. Appl Environ Microbiol. 2017. PMID: 27913420 Free PMC article.
-
Breeding for Resistance to Fusarium Wilt of Tomato: A Review.Genes (Basel). 2021 Oct 23;12(11):1673. doi: 10.3390/genes12111673. Genes (Basel). 2021. PMID: 34828278 Free PMC article. Review.
-
The arms race between tomato and Fusarium oxysporum.Mol Plant Pathol. 2010 Mar;11(2):309-14. doi: 10.1111/j.1364-3703.2009.00605.x. Mol Plant Pathol. 2010. PMID: 20447279 Free PMC article. Review.
Cited by
-
FolVps9, a Guanine Nucleotide Exchange Factor for FolVps21, Is Essential for Fungal Development and Pathogenicity in Fusarium oxysporum f. sp. lycopersici.Front Microbiol. 2019 Nov 14;10:2658. doi: 10.3389/fmicb.2019.02658. eCollection 2019. Front Microbiol. 2019. PMID: 31798569 Free PMC article.
-
Transcriptional responses to Fusarium oxysporum f. sp. lycopersici (Sacc.) Snyder & Hansen infection in three Colombian tomato cultivars.BMC Plant Biol. 2021 Sep 8;21(1):412. doi: 10.1186/s12870-021-03187-z. BMC Plant Biol. 2021. PMID: 34496757 Free PMC article.
-
Xylem Sap Proteomics Reveals Distinct Differences Between R Gene- and Endophyte-Mediated Resistance Against Fusarium Wilt Disease in Tomato.Front Microbiol. 2018 Dec 4;9:2977. doi: 10.3389/fmicb.2018.02977. eCollection 2018. Front Microbiol. 2018. PMID: 30564219 Free PMC article.
-
The Role of Pathogen-Secreted Proteins in Fungal Vascular Wilt Diseases.Int J Mol Sci. 2015 Oct 9;16(10):23970-93. doi: 10.3390/ijms161023970. Int J Mol Sci. 2015. PMID: 26473835 Free PMC article. Review.
-
Fungal effectors, the double edge sword of phytopathogens.Curr Genet. 2021 Feb;67(1):27-40. doi: 10.1007/s00294-020-01118-3. Epub 2020 Nov 4. Curr Genet. 2021. PMID: 33146780 Review.
References
-
- Alexander, L.J. and Tucker, C.M. (1945) Physiological specialization in the Tomato wilt fungus Fusarium oxysporum f. sp. lycopersici . J. Agric. Res. 70, 303–313.
-
- Bi, G. , Liebrand, T.W.H. , Bye, R.R. , Postma, J. , van der Burgh, A.M. , Robatzek, S. , Xu, X. and Joosten, M.H.A.J. (2015) SOBIR1 requires the GxxxG dimerization motif in its transmembrane domain to form constitutive complexes with receptor‐like proteins. Mol. Plant Pathol. doi: 10.1111/mpp.12266. - DOI - PMC - PubMed
-
- Bohn, G. and Tucker, C. (1939) Immunity to Fusarium wilt in the tomato. Science, 89, 603–604. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

