Redox-modulating agents target NOX2-dependent IKKε oncogenic kinase expression and proliferation in human breast cancer cell lines
- PMID: 26177467
- PMCID: PMC4511630
- DOI: 10.1016/j.redox.2015.06.010
Redox-modulating agents target NOX2-dependent IKKε oncogenic kinase expression and proliferation in human breast cancer cell lines
Abstract
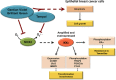
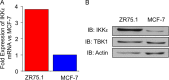
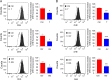
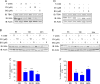
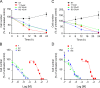
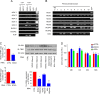
Oxidative stress is considered a causative factor in carcinogenesis, but also in the development of resistance to current chemotherapies. The appropriate usage of redox-modulating compounds is limited by the lack of knowledge of their impact on specific molecular pathways. Increased levels of the IKKε kinase, as a result of gene amplification or aberrant expression, are observed in a substantial number of breast carcinomas. IKKε not only plays a key role in cell transformation and invasiveness, but also in the development of resistance to tamoxifen. Here, we studied the effect of in vitro treatment with the redox-modulating triphenylmethane dyes, Gentian Violet and Brilliant Green, and nitroxide Tempol on IKKε expression and cell proliferation in the human breast cancer epithelial cell lines exhibiting amplification of IKKε, MCF-7 and ZR75.1. We show that Gentian Violet, Brilliant Green and Tempol significantly decrease intracellular superoxide anion levels and inhibit IKKε expression and cell viability. Treatment with Gentian Violet and Brilliant Green was associated with a reduced cyclin D1 expression and activation of caspase 3 and/or 7. Tempol decreased cyclin D1 expression in both cell lines, while activation of caspase 7 was only observed in MCF-7 cells. Silencing of the superoxide-generating NOX2 NADPH oxidase expressed in breast cancer cells resulted in the significant reduction of IKKε expression. Taken together, our results suggest that redox-modulating compounds targeting NOX2 could present a particular therapeutic interest in combination therapy against breast carcinomas exhibiting IKKε amplification.
Keywords: Breast cancer; Cell growth; IKKε; NADPH oxidase; Tempol; Triphenylmethane dyes.
Copyright © 2015 The Authors. Published by Elsevier B.V. All rights reserved.
Figures

Similar articles
-
Regulation of pancreatic cancer growth by superoxide.Mol Carcinog. 2013 Jul;52(7):555-67. doi: 10.1002/mc.21891. Epub 2012 Mar 5. Mol Carcinog. 2013. PMID: 22392697 Free PMC article.
-
Silencing of the IKKε gene by siRNA inhibits invasiveness and growth of breast cancer cells.Breast Cancer Res. 2010;12(5):R74. doi: 10.1186/bcr2644. Epub 2010 Sep 23. Breast Cancer Res. 2010. PMID: 20863366 Free PMC article.
-
ROS-induced ROS release orchestrated by Nox4, Nox2, and mitochondria in VEGF signaling and angiogenesis.Am J Physiol Cell Physiol. 2017 Jun 1;312(6):C749-C764. doi: 10.1152/ajpcell.00346.2016. Epub 2017 Apr 19. Am J Physiol Cell Physiol. 2017. PMID: 28424170 Free PMC article.
-
NADPH Oxidase-2 and Atherothrombosis: Insight From Chronic Granulomatous Disease.Arterioscler Thromb Vasc Biol. 2017 Feb;37(2):218-225. doi: 10.1161/ATVBAHA.116.308351. Epub 2016 Dec 8. Arterioscler Thromb Vasc Biol. 2017. PMID: 27932349 Review.
-
[Superoxide-generating enzymes NADPH oxidases, potential targets of drug therapy: various mechanisms for regulation of their expression].Nihon Yakurigaku Zasshi. 2013 Dec;142(6):285-90. doi: 10.1254/fpj.142.285. Nihon Yakurigaku Zasshi. 2013. PMID: 24334927 Review. Japanese. No abstract available.
Cited by
-
Analysis of the Effect of Gentian Violet on Apoptosis and Proliferation in Cutaneous T-Cell Lymphoma in an In Vitro Study.JAMA Dermatol. 2018 Oct 1;154(10):1191-1198. doi: 10.1001/jamadermatol.2018.2756. JAMA Dermatol. 2018. PMID: 30167641 Free PMC article.
-
Dihydrotanshinone-Induced NOX5 Activation Inhibits Breast Cancer Stem Cell through the ROS/Stat3 Signaling Pathway.Oxid Med Cell Longev. 2019 Mar 24;2019:9296439. doi: 10.1155/2019/9296439. eCollection 2019. Oxid Med Cell Longev. 2019. PMID: 31019654 Free PMC article.
-
The Association between NADPH Oxidase 2 (NOX2) and Drug Resistance in Cancer.Curr Cancer Drug Targets. 2024;24(12):1195-1212. doi: 10.2174/0115680096277328240110062433. Curr Cancer Drug Targets. 2024. PMID: 38362697 Review.
-
Paradoxical roles of dual oxidases in cancer biology.Free Radic Biol Med. 2017 Sep;110:117-132. doi: 10.1016/j.freeradbiomed.2017.05.024. Epub 2017 May 31. Free Radic Biol Med. 2017. PMID: 28578013 Free PMC article. Review.
-
Albendazole as a promising molecule for tumor control.Redox Biol. 2016 Dec;10:90-99. doi: 10.1016/j.redox.2016.09.013. Epub 2016 Sep 22. Redox Biol. 2016. PMID: 27710854 Free PMC article.
References
-
- Thiery J.P., Sastre-Garau X., Vincent-Salomon B., Sigal-Zafrani X., Pierga J.Y., Decraene C., Meyniel J.P., Gravier E., Asselain B., De Rycke Y., Hupe P., Barillot E., Ajaz S., Faraldo M., Deugnier M.A., Glukhova M., Medina D., Breast Cancer Group Challenges in the stratification of breast tumors for tailored therapies. Bull. Cancer. 2006;93(8):E81–E89. 16935776 - PubMed
-
- Eddy S.F., Guo S., Demicco E.G., Romieu-Mourez R., Landesman-Bollag E., Seldin D.C., Sonenshein G.E. Inducible IkappaB kinase/IkappaB kinase epsilon expression is induced by CK2 and promotes aberrant nuclear factor-kappaB activation in breast cancer cells. Cancer Res. 2005;65(24):11375–11383. 16357145 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

