Effect of Introducing Xpert MTB/RIF to Test and Treat Individuals at Risk of Multidrug-Resistant Tuberculosis in Kazakhstan: A Prospective Cohort Study
- PMID: 26181578
- PMCID: PMC4504513
- DOI: 10.1371/journal.pone.0132514
Effect of Introducing Xpert MTB/RIF to Test and Treat Individuals at Risk of Multidrug-Resistant Tuberculosis in Kazakhstan: A Prospective Cohort Study
Erratum in
-
Correction: Effect of Introducing Xpert MTB/RIF to Test and Treat Individuals at Risk of Multidrug-Resistant Tuberculosis in Kazakhstan: A Prospective Cohort Study.PLoS One. 2015 Aug 19;10(8):e0136368. doi: 10.1371/journal.pone.0136368. eCollection 2015. PLoS One. 2015. PMID: 26287484 Free PMC article. No abstract available.
Abstract
Background: Xpert MTB/RIF (Xpert) was piloted in Kazakhstan to detect tuberculosis (TB) and rifampicin resistance (RR-)TB among individuals at risk of multidrug-resistant (MDR-) TB. This study assessed the performance of Xpert compared to conventional diagnostic methods, RR-TB case detection among various risk groups, treatment initiation and time to diagnosis and treatment.
Methods: Eligible individuals were tested with Xpert, smear microscopy, culture and drug-susceptibility testing (DST) at the national TB reference laboratory and three provincial laboratories. Data was collected prospectively from August 2012 to May 2013 from routine laboratory and treatment registers.
Results: A total of 5,611 Xpert tests were performed mostly targeting contacts of MDR-TB patients, 'other' presumptive MDR-TB patients, and retreatment cases (26%, 24% and 22%, respectively). Compared to phenotypic DST, the positive predictive value of Xpert to detect RR-TB was 93.1% and 96.4% and the negative predictive value was 94.6% and 92.7% using solid and liquid culture media, respectively. RR-TB detection was highest among (former) prisoners, retreatment cases, people living with HIV/AIDS (PLWHA), and TB patients with positive smears after intensive phase of treatment (59%, 58%, 54% and 53% among TB positives, respectively). 88.9% of RR-TB patients were registered to have started second-line TB treatment. Median time to diagnosis with Xpert was 0.0 days (IQR 0.0-1.0), time from diagnosis to start of first-line treatment 3.0 days (IQR 1.0-7.0), and to start of second-line treatment 7.0 days (IQR 4.0-16).
Conclusions: Compared to conventional culture and DST, Xpert had a shorter result turn-around-time and excellent concordance to detect RR-TB. Time from sputum collection to start of second-line treatment was reduced to one week. The yield of Xpert could be maximized by increasing referrals from penitentiary and HIV centers to TB centers.
Conflict of interest statement
Figures
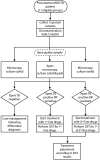
References
-
- World Health Organization. Global tuberculosis report 2013. Geneva: WHO; 2013.
-
- World Health Organization. Tuberculosis country work summary: Kazakhstan. WHO Regional Office for Europe; 2011. Available: http://www.euro.who.int/en/health-topics/communicable-diseases/tuberculo.... Accessed 8 Nov 2014.
-
- USAID. Treatment for TB moves from hospital to home in Kazakhstan. Available: http://www.usaid.gov/results-data/success-stories/new-patient-centered-m.... Accessed 4 Dec 2014.
-
- World Health Organization; Accelerating the implementation of collaborative TB/HIV activities in the WHO European Region, 16–17 July, 2010. Vienna: WHO and Stop TB Partnership TB/HIV Working Group; 2010. WHO_HTM_TB_2010.9.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

