Our Fat Future: Translating Adipose Stem Cell Therapy
- PMID: 26185256
- PMCID: PMC4542878
- DOI: 10.5966/sctm.2015-0071
Our Fat Future: Translating Adipose Stem Cell Therapy
Abstract
Human adipose stem cells (hASCs) have the potential to treat patients with a variety of clinical conditions. Recent advancements in translational research, regulatory policy, and industry have positioned hASCs on the threshold of clinical translation. We discuss the progress and challenges of bringing adipose stem cell therapy into mainstream clinical use.
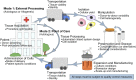
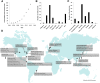
Significance: This article details the advances made in recent years that have helped move human adipose stem cell therapy toward mainstream clinical use from a translational research, regulatory policy, and industrial standpoint. Four recurrent themes in translational technology as they pertain to human adipose stem cells are discussed: automated closed-system operations, biosensors and real-time monitoring, biomimetics, and rapid manufacturing. In light of recent FDA guidance documents, regulatory concerns about adipose stem cell therapy are discussed. Finally, an update is provided on the current state of clinical trials and the emerging industry that uses human adipose stem cells. This article is expected to stimulate future studies in translational adipose stem cell research.
©AlphaMed Press.
Figures
References
-
- Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315–317. - PubMed
-
- Bourin P, Bunnell BA, Casteilla L, et al. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: A joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT) Cytotherapy. 2013;15:641–648. - PMC - PubMed
-
- Gir P, Oni G, Brown SA, et al. Human adipose stem cells: Current clinical applications. Plast Reconstr Surg. 2012;129:1277–1290. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

