MEK2 is a prognostic marker and potential chemo-sensitizing target for glioma patients undergoing temozolomide treatment
- PMID: 26189368
- PMCID: PMC5037281
- DOI: 10.1038/cmi.2015.46
MEK2 is a prognostic marker and potential chemo-sensitizing target for glioma patients undergoing temozolomide treatment
Abstract
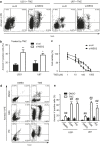
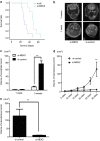
Although temozolomide (TMZ) is the first-line chemotherapeutic agent for glioblastoma, it is often non-curative due to drug resistance. To overcome the resistance of glioblastoma cells to TMZ, it is imperative to identify prognostic markers for outcome prediction and to develop chemo-sensitizing agents. Here, the gene expression profiles of TMZ-resistant and TMZ-sensitive samples were compared by microarray analysis, and mitogen-activated protein kinase kinase 2 (MEK2) was upregulated specifically in resistant glioma cells but not in sensitive tumor cells or non-tumor tissues. Moreover, a comprehensive analysis of patient data revealed that the increased level of MEK2 expression correlated well with the advancement of glioma grade and worse prognosis in response to TMZ treatment. Furthermore, reducing the level of MEK2 in U251 glioma cell lines or xenografted glioma models through shRNA-mediated gene knockdown inhibited cell proliferation and enhanced the sensitivity of cells toward TMZ treatment. Further analysis of tumor samples from glioma patients by real-time PCR indicated that an increased MEK2 expression level was closely associated with the activation of many drug resistance genes. Finally, these resistance genes were downregulated after MEK2 was silenced in vitro, suggesting that the mechanism of MEK2-induced chemo-resistance could be mediated by the transcriptional activation of these resistance genes. Collectively, our data indicated that the expression level of MEK2 could serve as a prognostic marker for glioma chemotherapy and that MEK2 antagonists can be used as chemo-sensitizers to enhance the treatment efficacy of TMZ.
Figures




References
-
- Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 2005; 352: 987–996. - PubMed
-
- Corsa P, Parisi S, Raguso A, Troiano M, Perrone A, Cossa S et al. Temozolomide and radiotherapy as first-line treatment of high-grade gliomas. Tumori 2006; 92: 299–305. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

