Stereospecific nickel-catalyzed cross-coupling reactions of benzylic ethers and esters
- PMID: 26197033
- PMCID: PMC4956245
- DOI: 10.1021/acs.accounts.5b00223
Stereospecific nickel-catalyzed cross-coupling reactions of benzylic ethers and esters
Abstract
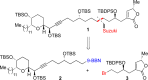
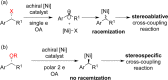
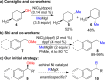
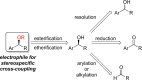
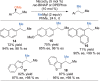
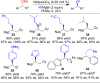
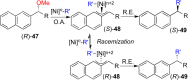
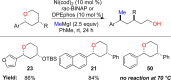
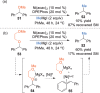
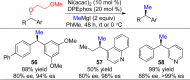
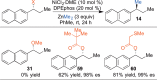
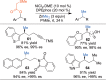
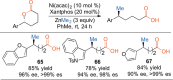
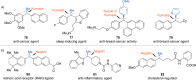
This Account presents the development of a suite of stereospecific alkyl-alkyl cross-coupling reactions employing nickel catalysts. Our reactions complement related nickel-catalyzed stereoconvergent cross-coupling reactions from a stereochemical and mechanistic perspective. Most reactions of alkyl electrophiles with low-valent nickel complexes proceed through alkyl radicals and thus are stereoablative; the correct enantioselective catalyst can favor the formation of one enantiomer. Our reactions, in contrast, are stereospecific. Enantioenriched ethers and esters are cleanly converted to cross-coupled products with high stereochemical fidelity. While mechanistic details are still to be refined, our results are consistent with a polar, two-electron oxidative addition that avoids the formation of radical intermediates. This reactivity is unusual for a first-row transition metal. The cross-coupling reactions engage a range of benzylic ethers and esters, including methyl ethers, tetrahydropyrans, tetrahydrofurans, esters, and lactones. Coordination of the arene substituent to the nickel catalyst accelerates the reactions. Arenes with low aromatic stabilization energies, such as naphthalene, benzothiophene, and furan, serve as the best ligands and provide the highest reactivity. Traceless directing groups that accelerate reactions of sluggish substrates are described, providing partial compensation for arene coordination. Kumada, Negishi, and Suzuki reactions provide incorporation of a broad range of transmetalating agents. In Kumada coupling reactions, a full complement of Grigard reagents, including methyl, n-alkyl, and aryl Grignard reagents, are employed. In reactions employing methylmagnesium iodide, ligation of the nickel catalyst by rac-BINAP or DPEphos provides the highest yield and stereospecificity. For all other Grignard reagents, Ni(dppe)Cl2 has emerged as the best catalyst. Negishi cross-coupling reactions employing dimethylzinc are reported as a strategy to increase the functional group tolerance of the reaction. We also describe Suzuki reactions using arylboronic esters. These reactions provided the first example in the series of a switch in stereochemical outcome. The reactions maintain stereospecificity, but reactions employing different achiral ligands provide opposite enantiomers of the product. Use of an N-heterocyclic carbene ligand, SIMes, provides inversion, consistent with our prior work in Kumada and Negishi coupling reactions. Use of the electron-rich phosphine PCy3, however, provides retention with stereospecificity, signaling a change in the mechanistic details. Potential applications of the reported cross-coupling reactions include the synthesis of medicinal agents containing the 2-arylalkane and 1,1-diarylalkane moieties, which are pharmacophores in medicinal chemistry. These moieties are found in compounds with activity against a broad range of indications, including cancer, heart disease, diabetes, osteoporosis, smallpox, tuberculosis, and insomnia. We highlight representative examples of bioactive compounds that we have prepared with high enantioselectivity employing our methods, as well as the discovery of a new anti-cancer agent.
Conflict of interest statement
The authors declare no competing financial interest.
Figures






References
-
-
For a transition-metal-free strategy, including the formation of quaternary stereocenters, see:
- Balieu S.; Hallett G. E.; Burns M.; Bootwicha T.; Studley J.; Aggarwal V. Toward Ideality: The Synthesis of (+)-Kalkitoxin and (+)-Hydroxyphthioceranic Acid by Assembly-Line Synthesis. J. Am. Chem. Soc. 2015, 137, 4398–4403. 10.1021/ja512875g. - DOI - PubMed
- Bonet A.; Odachowski M.; Leonori D.; Essafi S.; Aggarwal V. K. Enantiospecific sp2-sp3 Coupling of Secondary and Tertiary Boronic Esters. Nat. Chem. 2014, 6, 584–589. 10.1038/nchem.1971. - DOI - PubMed
-
-
- Nicolaou K. C.; Sorensen E. J.. Classics in Total Synthesis; Wiley-VCH: Weinheim, Germany, 1996; p 185.
- Carreira E. M.; Kvaerno L.. Classics in Stereoselective Synthesis; Wiley-VCH: Weinheim, Germany, 2009.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

