Neuroprotective Effects of the Glutamate Transporter Activator (R)-(-)-5-methyl-1-nicotinoyl-2-pyrazoline (MS-153) following Traumatic Brain Injury in the Adult Rat
- PMID: 26200170
- PMCID: PMC4892232
- DOI: 10.1089/neu.2015.4079
Neuroprotective Effects of the Glutamate Transporter Activator (R)-(-)-5-methyl-1-nicotinoyl-2-pyrazoline (MS-153) following Traumatic Brain Injury in the Adult Rat
Abstract
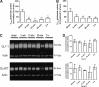
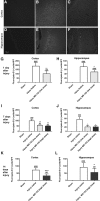
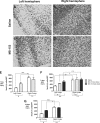
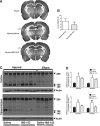
Traumatic brain injury (TBI) in humans and in animals leads to an acute and sustained increase in tissue glutamate concentrations within the brain, triggering glutamate-mediated excitotoxicity. Excitatory amino acid transporters (EAATs) are responsible for maintaining extracellular central nervous system glutamate concentrations below neurotoxic levels. Our results demonstrate that as early as 5 min and up to 2 h following brain trauma in brain-injured rats, the activity (Vmax) of EAAT2 in the cortex and the hippocampus was significantly decreased, compared with sham-injured animals. The affinity for glutamate (KM) and the expression of glutamate transporter 1 (GLT-1) and glutamate aspartate transporter (GLAST) were not altered by the injury. Administration of (R)-(-)-5-methyl-1-nicotinoyl-2-pyrazoline (MS-153), a GLT-1 activator, beginning immediately after injury and continuing for 24 h, significantly decreased neurodegeneration, loss of microtubule-associated protein 2 and NeuN (+) immunoreactivities, and attenuated calpain activation in both the cortex and the hippocampus at 24 h after the injury; the reduction in neurodegeneration remained evident up to 14 days post-injury. In synaptosomal uptake assays, MS-153 up-regulated GLT-1 activity in the naïve rat brain but did not reverse the reduced activity of GLT-1 in traumatically-injured brains. This study demonstrates that administration of MS-153 in the acute post-traumatic period provides acute and long-term neuroprotection for TBI and suggests that the neuroprotective effects of MS-153 are related to mechanisms other than GLT-1 activation, such as the inhibition of voltage-gated calcium channels.
Keywords: EAAT2/GLT-1; MS-153; glutamate uptake; neuroprotection; traumatic brain injury.
Figures

Similar articles
-
Early loss of the glutamate transporter splice-variant GLT-1v in rat cerebral cortex following lateral fluid-percussion injury.Glia. 2005 Jan 1;49(1):121-33. doi: 10.1002/glia.20099. Glia. 2005. PMID: 15390098
-
The role of astrocytic glutamate transporters GLT-1 and GLAST in neurological disorders: Potential targets for neurotherapeutics.Neuropharmacology. 2019 Dec 15;161:107559. doi: 10.1016/j.neuropharm.2019.03.002. Epub 2019 Mar 6. Neuropharmacology. 2019. PMID: 30851309 Free PMC article. Review.
-
Histamine up-regulates astrocytic glutamate transporter 1 and protects neurons against ischemic injury.Neuropharmacology. 2014 Feb;77:156-66. doi: 10.1016/j.neuropharm.2013.06.012. Epub 2013 Jun 18. Neuropharmacology. 2014. PMID: 23791559
-
Expression of glutamate transporters GLT-1 and GLAST in different regions of rat brain during the course of experimental autoimmune encephalomyelitis.Neuroscience. 2008 Jul 31;155(1):45-52. doi: 10.1016/j.neuroscience.2008.05.025. Epub 2008 May 24. Neuroscience. 2008. PMID: 18572325
-
Current approaches to enhance glutamate transporter function and expression.J Neurochem. 2015 Sep;134(6):982-1007. doi: 10.1111/jnc.13200. Epub 2015 Aug 14. J Neurochem. 2015. PMID: 26096891 Review.
Cited by
-
Revisiting Traumatic Brain Injury: From Molecular Mechanisms to Therapeutic Interventions.Biomedicines. 2020 Sep 29;8(10):389. doi: 10.3390/biomedicines8100389. Biomedicines. 2020. PMID: 33003373 Free PMC article. Review.
-
Incretin Mimetics as Rational Candidates for the Treatment of Traumatic Brain Injury.ACS Pharmacol Transl Sci. 2019 Apr 12;2(2):66-91. doi: 10.1021/acsptsci.9b00003. Epub 2019 Feb 11. ACS Pharmacol Transl Sci. 2019. PMID: 31396586 Free PMC article.
-
Glutamatergic systems in neuropathic pain and emerging non-opioid therapies.Pharmacol Res. 2022 Nov;185:106492. doi: 10.1016/j.phrs.2022.106492. Epub 2022 Oct 10. Pharmacol Res. 2022. PMID: 36228868 Free PMC article. Review.
-
Assessment of neuropharmacological potential of low molecular weight components extracted from Rhinella schneideri toad poison.J Venom Anim Toxins Incl Trop Dis. 2019 Apr 18;25:e148418. doi: 10.1590/1678-9199-JVATITD-1484-18. eCollection 2019. J Venom Anim Toxins Incl Trop Dis. 2019. PMID: 31131006 Free PMC article.
-
Mechanisms of Endogenous Neuroprotective Effects of Astrocytes in Brain Injury.Oxid Med Cell Longev. 2018 Apr 1;2018:6501031. doi: 10.1155/2018/6501031. eCollection 2018. Oxid Med Cell Longev. 2018. PMID: 29805731 Free PMC article. Review.
References
-
- Janowitz T. and Menon D.K. (2010). Exploring new routes for neuroprotective drug development in traumatic brain injury. Sci. Transl. Med. 2, 21–27 - PubMed
-
- Palmer A.M., Marion D.W., Botscheller M.L., Swedlow P.E., Styren S.D., and DeKosky S.T. (1993). Traumatic brain injury-induced excitotoxicity assessed in a controlled cortical impact model. J. Neurochem. 61, 2015–2024 - PubMed
-
- Brown J.I., Baker A.J., Konasiewicz S.J., and Moulton R.J. (1998). Clinical significance of CSF glutamate concentrations following severe traumatic brain injury in humans. J. Neurotrauma 15, 253–263 - PubMed
-
- Vespa P., Prins M., Ronne-Engstrom E., Caron M., Shalmon E., Hovda D.A., Martin N.A., and Becker D.P. (1998). Increase in extracellular glutamate caused by reduced cerebral perfusion pressure and seizures after human traumatic brain injury: a microdialysis study. J. Neurosurg. 89, 971–982 - PubMed
-
- Baker A.J., Moulton R.J., MacMillan V.H., and Shedden P.M. (1993). Excitatory amino acids in cerebrospinal fluid following traumatic brain injury in humans. J. Neurosurg. 79, 369–372 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

