Identification and Characterization of a Novel Family of Cysteine-Rich Peptides (MgCRP-I) from Mytilus galloprovincialis
- PMID: 26201648
- PMCID: PMC4558851
- DOI: 10.1093/gbe/evv133
Identification and Characterization of a Novel Family of Cysteine-Rich Peptides (MgCRP-I) from Mytilus galloprovincialis
Abstract
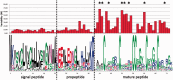
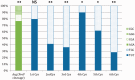
We report the identification of a novel gene family (named MgCRP-I) encoding short secreted cysteine-rich peptides in the Mediterranean mussel Mytilus galloprovincialis. These peptides display a highly conserved pre-pro region and a hypervariable mature peptide comprising six invariant cysteine residues arranged in three intramolecular disulfide bridges. Although their cysteine pattern is similar to cysteines-rich neurotoxic peptides of distantly related protostomes such as cone snails and arachnids, the different organization of the disulfide bridges observed in synthetic peptides and phylogenetic analyses revealed MgCRP-I as a novel protein family. Genome- and transcriptome-wide searches for orthologous sequences in other bivalve species indicated the unique presence of this gene family in Mytilus spp. Like many antimicrobial peptides and neurotoxins, MgCRP-I peptides are produced as pre-propeptides, usually have a net positive charge and likely derive from similar evolutionary mechanisms, that is, gene duplication and positive selection within the mature peptide region; however, synthetic MgCRP-I peptides did not display significant toxicity in cultured mammalian cells, insecticidal, antimicrobial, or antifungal activities. The functional role of MgCRP-I peptides in mussel physiology still remains puzzling.
Keywords: antimicrobial peptide; bivalve mollusk; mussel; toxin; transcriptome.
© The Author(s) 2015. Published by Oxford University Press on behalf of the Society for Molecular Biology and Evolution.
Figures



Similar articles
-
Comparative Genomics Reveals 13 Different Isoforms of Mytimycins (A-M) in Mytilus galloprovincialis.Int J Mol Sci. 2021 Mar 22;22(6):3235. doi: 10.3390/ijms22063235. Int J Mol Sci. 2021. PMID: 33810127 Free PMC article.
-
Big defensins and mytimacins, new AMP families of the Mediterranean mussel Mytilus galloprovincialis.Dev Comp Immunol. 2012 Feb;36(2):390-9. doi: 10.1016/j.dci.2011.08.003. Epub 2011 Aug 18. Dev Comp Immunol. 2012. PMID: 21871485
-
Myticin, a novel cysteine-rich antimicrobial peptide isolated from haemocytes and plasma of the mussel Mytilus galloprovincialis.Eur J Biochem. 1999 Oct 1;265(1):71-8. doi: 10.1046/j.1432-1327.1999.00654.x. Eur J Biochem. 1999. PMID: 10491159
-
Genomics and immunity of the Mediterranean mussel Mytilus galloprovincialis in a changing environment.Fish Shellfish Immunol. 2019 Jul;90:440-445. doi: 10.1016/j.fsi.2019.04.064. Epub 2019 Apr 30. Fish Shellfish Immunol. 2019. PMID: 31048040 Review.
-
Cysteine-rich mini-proteins in human biology.Curr Top Med Chem. 2012;12(14):1514-33. doi: 10.2174/156802612802652411. Curr Top Med Chem. 2012. PMID: 22827521 Review.
Cited by
-
Unveiling the Impact of Gene Presence/Absence Variation in Driving Inter-Individual Sequence Diversity within the CRP-I Gene Family in Mytilus spp.Genes (Basel). 2023 Mar 24;14(4):787. doi: 10.3390/genes14040787. Genes (Basel). 2023. PMID: 37107545 Free PMC article.
-
Myticalins: A Novel Multigenic Family of Linear, Cationic Antimicrobial Peptides from Marine Mussels (Mytilus spp.).Mar Drugs. 2017 Aug 22;15(8):261. doi: 10.3390/md15080261. Mar Drugs. 2017. PMID: 28829401 Free PMC article.
-
The purplish bifurcate mussel Mytilisepta virgata gene expression atlas reveals a remarkable tissue functional specialization.BMC Genomics. 2017 Aug 8;18(1):590. doi: 10.1186/s12864-017-4012-z. BMC Genomics. 2017. PMID: 28789640 Free PMC article.
-
Venom Diversity and Evolution in the Most Divergent Cone Snail Genus Profundiconus.Toxins (Basel). 2019 Oct 28;11(11):623. doi: 10.3390/toxins11110623. Toxins (Basel). 2019. PMID: 31661832 Free PMC article.
-
Parallel identification of novel antimicrobial peptide sequences from multiple anuran species by targeted DNA sequencing.BMC Genomics. 2018 Nov 20;19(1):827. doi: 10.1186/s12864-018-5225-5. BMC Genomics. 2018. PMID: 30458708 Free PMC article.
References
-
- Adams DJ, Alewood PF, Craik DJ, Drinkwater RD, Lewis RJ. 1999. Conotoxins and their potential pharmaceutical applications. Drug Dev Res. 46:219–234.
-
- Aguilar MB, de la Rosa RAC, Falcón A, Olivera BM, Heimer de la Cotera EP. 2009. Peptide pal9a from the venom of the turrid snail Polystira albida from the Gulf of Mexico: purification, characterization, and comparison with P-conotoxin-like (framework IX) conoidean peptides. Peptides 30:467–476 - PMC - PubMed
-
- Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. 1990. Basic local alignment search tool. J Mol Biol. 215:403–410. - PubMed
-
- Bartlett TC, et al. 2002. Crustins, homologues of an 11.5-kDa antibacterial peptide, from two species of penaeid shrimp, Litopenaeus vannamei and Litopenaeus setiferus. Mar Biotechnol. 4:278–293. - PubMed
-
- Bassim S, Genard B, Gauthier-Clerc S, Moraga D, Tremblay R. 2014. Ontogeny of bivalve immunity: assessing the potential of next-generation sequencing techniques. Rev Aquac. 6:1–21.
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

