Obif, a Transmembrane Protein, Is Required for Bone Mineralization and Spermatogenesis in Mice
- PMID: 26207632
- PMCID: PMC4514473
- DOI: 10.1371/journal.pone.0133704
Obif, a Transmembrane Protein, Is Required for Bone Mineralization and Spermatogenesis in Mice
Abstract
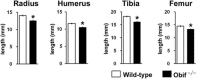
Background: Various kinds of transmembrane and secreted proteins play pivotal roles in development through cell-cell communication. We previously reported that Obif (Osteoblast induction factor, Tmem119), encoding a single transmembrane protein, is expressed in differentiating osteoblasts, and that Obif-/- mice exhibit significantly reduced bone volume in the femur. In the current study, we characterized the Obif protein and further investigated the biological phenotypes of a variety of tissues in Obif-/- mice.
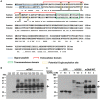
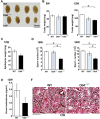
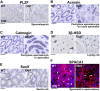
Results: First, we found that O-glycosylation of the Obif protein occurs at serine residue 36 in the Obif extracellular domain. Next, we observed that Obif-/- mice exhibit bone dysplasia in association with significantly increased osteoid volume per osteoid surface (OV/OS) and osteoid maturation time (Omt), and significantly decreased mineral apposition rate (MAR) and bone formation rate per bone surface (BFR/BS). In addition, we observed that Obif-/- mice show a significant decrease in testis weight as well as in sperm number. By histological analysis, we found that Obif is expressed in spermatocytes and spermatids in the developing testis and that spermatogenesis is halted at the round spermatid stage in the Obif-/- testis that lacks sperm. However, the number of litters fathered by male mice was slightly reduced in Obif-/- mice compared with wild-type mice, although this was not statistically significant.
Conclusions: Our results, taken together with previous observations, indicate that Obif is a type Ia transmembrane protein whose N-terminal region is O-glycosylated. In addition, we found that Obif is required for normal bone mineralization and late testicular differentiation in vivo. These findings suggest that Obif plays essential roles in the development of multiple tissues.
Conflict of interest statement
Figures


Similar articles
-
OBIF, an osteoblast induction factor, plays an essential role in bone formation in association with osteoblastogenesis.Dev Growth Differ. 2012 May;54(4):474-80. doi: 10.1111/j.1440-169X.2012.01333.x. Epub 2012 Mar 15. Dev Growth Differ. 2012. PMID: 22416756
-
Isolation and characterization of a novel plasma membrane protein, osteoblast induction factor (obif), associated with osteoblast differentiation.BMC Dev Biol. 2009 Dec 21;9:70. doi: 10.1186/1471-213X-9-70. BMC Dev Biol. 2009. PMID: 20025746 Free PMC article.
-
Developmental expression pattern of a novel gene, TSG23/Tsg23, suggests a role in spermatogenesis.Mol Hum Reprod. 2009 Apr;15(4):223-30. doi: 10.1093/molehr/gap015. Epub 2009 Feb 24. Mol Hum Reprod. 2009. PMID: 19240080
-
Characterization and expression of mouse Cdc50c during spermatogenesis.Acta Biochim Biophys Sin (Shanghai). 2007 Oct;39(10):739-44. doi: 10.1111/j.1745-7270.2007.00342.x. Acta Biochim Biophys Sin (Shanghai). 2007. PMID: 17928922
-
Mice lacking the UBC4-testis gene have a delay in postnatal testis development but normal spermatogenesis and fertility.Mol Cell Biol. 2005 Aug;25(15):6346-54. doi: 10.1128/MCB.25.15.6346-6354.2005. Mol Cell Biol. 2005. PMID: 16024774 Free PMC article.
Cited by
-
CEP128 is involved in spermatogenesis in humans and mice.Nat Commun. 2022 Mar 16;13(1):1395. doi: 10.1038/s41467-022-29109-7. Nat Commun. 2022. PMID: 35296684 Free PMC article.
-
Can quantifying morphology and TMEM119 expression distinguish between microglia and infiltrating macrophages after ischemic stroke and reperfusion in male and female mice?J Neuroinflammation. 2021 Feb 22;18(1):58. doi: 10.1186/s12974-021-02105-2. J Neuroinflammation. 2021. PMID: 33618737 Free PMC article.
-
TMEM119 facilitates ovarian cancer cell proliferation, invasion, and migration via the PDGFRB/PI3K/AKT signaling pathway.J Transl Med. 2021 Mar 17;19(1):111. doi: 10.1186/s12967-021-02781-x. J Transl Med. 2021. PMID: 33731124 Free PMC article.
-
Human CD19-Targeted Mouse T Cells Induce B Cell Aplasia and Toxicity in Human CD19 Transgenic Mice.Mol Ther. 2018 Jun 6;26(6):1423-1434. doi: 10.1016/j.ymthe.2018.04.006. Epub 2018 Apr 7. Mol Ther. 2018. PMID: 29735365 Free PMC article.
-
Selection for bull fertility: a review.Transl Anim Sci. 2019 Dec 2;4(1):423-441. doi: 10.1093/tas/txz174. eCollection 2020 Jan. Transl Anim Sci. 2019. PMID: 32705001 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

