Transcription Factor Runx2 Promotes Aortic Fibrosis and Stiffness in Type 2 Diabetes Mellitus
- PMID: 26208651
- PMCID: PMC4553105
- DOI: 10.1161/CIRCRESAHA.115.306341
Transcription Factor Runx2 Promotes Aortic Fibrosis and Stiffness in Type 2 Diabetes Mellitus
Abstract
Rationale: Accelerated arterial stiffening is a major complication of diabetes mellitus with no specific therapy available to date.
Objective: The present study investigates the role of the osteogenic transcription factor runt-related transcription factor 2 (Runx2) as a potential mediator and therapeutic target of aortic fibrosis and aortic stiffening in diabetes mellitus.
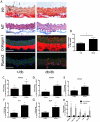
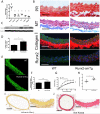
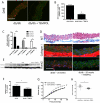
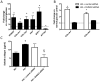
Methods and results: Using a murine model of type 2 diabetes mellitus (db/db mice), we identify progressive structural aortic stiffening that precedes the onset of arterial hypertension. At the same time, Runx2 is aberrantly upregulated in the medial layer of db/db aortae, as well as in thoracic aortic samples from patients with type 2 diabetes mellitus. Vascular smooth muscle cell-specific overexpression of Runx2 in transgenic mice increases expression of its target genes, Col1a1 and Col1a2, leading to medial fibrosis and aortic stiffening. Interestingly, increased Runx2 expression per se is not sufficient to induce aortic calcification. Using in vivo and in vitro approaches, we further demonstrate that expression of Runx2 in diabetes mellitus is regulated via a redox-sensitive pathway that involves a direct interaction of NF-κB with the Runx2 promoter.
Conclusions: In conclusion, this study highlights Runx2 as a previously unrecognized inducer of vascular fibrosis in the setting of diabetes mellitus, promoting arterial stiffness irrespective of calcification.
Keywords: arterial stiffness; diabetes mellitus; extracellular matrix; oxidative stress; smooth muscle cell.
© 2015 American Heart Association, Inc.
Figures

References
-
- Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27:1047–53. - PubMed
-
- Centers for Disease Control and Prevention . National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014. U.S. Department of Helath and Human Services; Atlanta, GA: 2014.
-
- Ryden L, Standl E, Bartnik M, Van den Berghe G, Betteridge J, de Boer MJ, Cosentino F, Jonsson B, Laakso M, Malmberg K, Priori S, Ostergren J, Tuomilehto J, Thrainsdottir I, Vanhorebeek I, Stramba-Badiale M, Lindgren P, Qiao Q, Priori SG, Blanc JJ, Budaj A, Camm J, Dean V, Deckers J, Dickstein K, Lekakis J, McGregor K, Metra M, Morais J, Osterspey A, Tamargo J, Zamorano JL, Deckers JW, Bertrand M, Charbonnel B, Erdmann E, Ferrannini E, Flyvbjerg A, Gohlke H, Juanatey JR, Graham I, Monteiro PF, Parhofer K, Pyorala K, Raz I, Schernthaner G, Volpe M, Wood D. Guidelines on diabetes, pre-diabetes, and cardiovascular diseases: executive summary. The Task Force on Diabetes and Cardiovascular Diseases of the European Society of Cardiology (ESC) and of the European Association for the Study of Diabetes (EASD) Eur Heart J. 2007;28:88–136. - PubMed
-
- Stehouwer C, Henry R, Ferreira I. Arterial stiffness in diabetes and the metabolic syndrome: a pathway to cardiovascular disease. Diabetologia. 2008;51:527–39. - PubMed
-
- Laurent S, Cockcroft J, Van Bortel L, Boutouyrie P, Giannattasio C, Hayoz D, Pannier B, Vlachopoulos C, Wilkinson I, Struijker-Boudier H. Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J. 2006;27:2588–605. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

