The female urinary microbiome in urgency urinary incontinence
- PMID: 26210757
- PMCID: PMC4556587
- DOI: 10.1016/j.ajog.2015.07.009
The female urinary microbiome in urgency urinary incontinence
Abstract
Objective: The purpose of this study was to characterize the urinary microbiota in women who are planning treatment for urgency urinary incontinence and to describe clinical associations with urinary symptoms, urinary tract infection, and treatment outcomes.
Study design: Catheterized urine samples were collected from multisite randomized trial participants who had no clinical evidence of urinary tract infection; 16S ribosomal RNA gene sequencing was used to dichotomize participants as either DNA sequence-positive or sequence-negative. Associations with demographics, urinary symptoms, urinary tract infection risk, and treatment outcomes were determined. In sequence-positive samples, microbiotas were characterized on the basis of their dominant microorganisms.
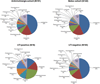
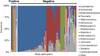
Results: More than one-half (51.1%; 93/182) of the participants' urine samples were sequence-positive. Sequence-positive participants were younger (55.8 vs 61.3 years old; P = .0007), had a higher body mass index (33.7 vs 30.1 kg/m(2); P = .0009), had a higher mean baseline daily urgency urinary incontinence episodes (5.7 vs 4.2 episodes; P < .0001), responded better to treatment (decrease in urgency urinary incontinence episodes, -4.4 vs -3.3; P = .0013), and were less likely to experience urinary tract infection (9% vs 27%; P = .0011). In sequence-positive samples, 8 major bacterial clusters were identified; 7 clusters were dominated not only by a single genus, most commonly Lactobacillus (45%) or Gardnerella (17%), but also by other taxa (25%). The remaining cluster had no dominant genus (13%).
Conclusion: DNA sequencing confirmed urinary bacterial DNA in many women with urgency urinary incontinence who had no signs of infection. Sequence status was associated with baseline urgency urinary incontinence episodes, treatment response, and posttreatment urinary tract infection risk.
Keywords: bacteria; microbiome; microbiota; urgency urinary incontinence; urinary tract infection.
Copyright © 2015 Elsevier Inc. All rights reserved.
Conflict of interest statement
Holly E. RICHTER, Ph.D., M.D.- Leadership: Society of Gynecologic Surgeons Board of Directors (no compensation); Health Publishing: Up-To-Date; Consultant: Pelvalon, Kimberly Clark; Meeting Lecturer: Symposia Medicus: Research Grants: Univ of CA/Pfizer, NIDDK, NICHD, NIAID, NIDDK/Yale, Pelvalon; Expert Review – Alabama Board of Medical Examiners
Anthony Visco, MD – Scientific Study/Trial: Pelvic Floor Disorders Network
Ingrid NYGAARD, M.D. – Health Publishing: Received an honorarium from Elsevier as Editor-in-Chief for AJOG.
Matthew D. BARBER, M.D., M.H.S – Scientific Study/Trial: Grants from NICHD, grants from foundation for Female Health Awareness, other from Elsevier, other from UptoDate, outside the submitted work.
Joseph Schaffer, MD – Health Publishing: McGraw-Hill Publishing – Royalties for six years; Meeting Participant/Lecturer: Astellas/GSK Pharmaceuticals; Scientific Study/Trial: Boston Scientific, Health Publishing: McGraw-Hill Publishing.
Pamela Moalli, MD, PhD – Scientific Study/Trial: Independent Research Agreement with ACell.
Rebecca ROGERS, MD – Health Publishing: McGraw Hill – Royalties from textbook.
Alan J. WOLFE, Ph.D. – Scientific Study/Trial: Investigator Initiated Grant from Astellas Pharmaceutical for a study outside the submitted work.
Linda BRUBAKER, MD – Scientific Study/Trial: Grants from NICHD (funding source for this study) and NIDDK during conduct of the study. Health Publishing: Personal fees from Up-To-Date.
The following authors report no conflict of interest: Dr. Meghan M. Pearce, Dr. Michael Zilliox, Ms. Krystal Thomas-White, Dr. Charles Nager, Dr. Vivian Sung, Dr. Ariana Smith, Dr. Tracy Nolen, Dr. Dennis Wallace, Dr. Susan Meikle and Dr. Xiaowu Gai.
Figures



References
-
- Brubaker L, Wolfe A. The new world of the urinary microbiome in women. Am J Obset Gynecol. In Press. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- U01 HD069031RT/HD/NICHD NIH HHS/United States
- U10-HD41249/HD/NICHD NIH HHS/United States
- U10 HD041261/HD/NICHD NIH HHS/United States
- U10 HD054215/HD/NICHD NIH HHS/United States
- U10 HD054214/HD/NICHD NIH HHS/United States
- 2-U10-HD041261-11/HD/NICHD NIH HHS/United States
- 2-U10-HD054215-06/HD/NICHD NIH HHS/United States
- 2-U10-HD054214-06/HD/NICHD NIH HHS/United States
- U10 HD054136/HD/NICHD NIH HHS/United States
- U10-HD054136/HD/NICHD NIH HHS/United States
- U10 HD054241/HD/NICHD NIH HHS/United States
- U10 HD041250/HD/NICHD NIH HHS/United States
- 1G20RR030939-0 1/RR/NCRR NIH HHS/United States
- U10 HD069025/HD/NICHD NIH HHS/United States
- 1-U10-HD069006-01/HD/NICHD NIH HHS/United States
- U01 HD041249/HD/NICHD NIH HHS/United States
- G20 RR030939/RR/NCRR NIH HHS/United States
- U01 HD069031/HD/NICHD NIH HHS/United States
- UG1 HD069010/HD/NICHD NIH HHS/United States
- U10 HD069013/HD/NICHD NIH HHS/United States
- 2-U10-HD04267-12/HD/NICHD NIH HHS/United States
- 2-U10-HD054241-06/HD/NICHD NIH HHS/United States
- 1-U01-HD069010-01/HD/NICHD NIH HHS/United States
- U10 HD069006/HD/NICHD NIH HHS/United States
- U10 HD069010/HD/NICHD NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

