Use of a force-sensing automated open field apparatus in a longitudinal study of multiple behavioral deficits in CAG140 Huntington's disease model mice
- PMID: 26210937
- PMCID: PMC4564309
- DOI: 10.1016/j.bbr.2015.07.036
Use of a force-sensing automated open field apparatus in a longitudinal study of multiple behavioral deficits in CAG140 Huntington's disease model mice
Abstract
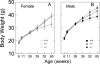
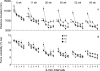
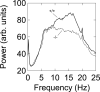
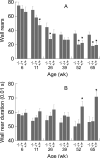
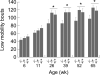
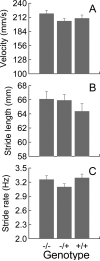
Behavioral testing of mouse models of Huntington's disease (HD) is a key component of preclinical assessment for potential pharmacological intervention. An open field with a force plate floor was used to quantify numerous spontaneous behaviors in a slowly progressing model of HD. CAG140 (+/+, +/-, -/-) male and female mice were compared in a longitudinal study from 6 to 65 weeks of age. Distance traveled, wall rears, wall rear duration, number of low mobility bouts, in-place movements, number of high velocity runs, and gait parameters (stride rate, stride length, and velocity) were extracted from the ground reaction forces recorded in 20-min actometer sessions. Beginning at 11 weeks, HD mice (both +/- and +/+) were consistently hypoactive throughout testing. Robust hypoactivity at 39 weeks of age was not accompanied by gait disturbances. By 52 and 65 weeks of age the duration of wall rears increased and in-place tremor-like movements emerged at 65 weeks of age in the +/+, but not in the +/- HD mice. Taken together, these results suggest that hypoactivity preceding frank motor dysfunction is a characteristic of CAG140 mice that may correspond to low motivation to move seen clinically in the premanifest/prediagnostic stage in human HD. The results also show that the force plate method provides a means for tracking the progression of behavioral dysfunction in HD mice beyond the stage when locomotion is lost while enabling quantification of tremor-like and similar in-place behaviors without a change in instrumentation. Use of force plate actometry also minimizes testing-induced enrichment effects when batteries of different tests are carried out longitudinally.
Keywords: CAG140 knock-in mouse; Distance traveled; Force plate; Gait; Huntington’s disease; Power spectra.
Copyright © 2015 Elsevier B.V. All rights reserved.
Figures

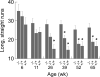
References
-
- The Huntington's Disease Collaborative Research Group A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington's disease chromosomes. Cell. 1993;72(6):971–83. PMID: 8458085. - PubMed
-
- Menalled L, Lutz C, Ramboz S, Brunner D, Lager B, Noble S, Park L, Howland D. A field guide to working with mouse models of Huntington's Disease. 2014 Chdifoundation.org.
-
- Menalled LB, Kudwa AE, Miller S, Fitzpatrick J, Watson-Johnson J, Keating N, Ruiz M, Mushlin R, Alosio W, McConnell K, Connor D, Murphy C, Oakeshott S, Kwan M, Beltran J, Ghavami A, Brunner D, Park LC, Ramboz S, Howland D. Comprehensive behavioral and molecular characterization of a new knock-in mouse model of Huntington's disease: zQ175. PLoS One. 2012;7(12):e49838. PMID: 23284626. - PMC - PubMed
-
- Fowler SC, Birkestrand BR, Chen R, Moss SJ, Vorontsova E, Wang G, Zarcone TJ. A force-plate actometer for quantitating rodent behaviors: illustrative data on locomotion, rotation, spatial patterning, stereotypies, and tremor. J Neurosci Methods. 2001;107(1-2):107–24. PMID: 11389948. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

