A novel laminin β gene BmLanB1-w regulates wing-specific cell adhesion in silkworm, Bombyx mori
- PMID: 26212529
- PMCID: PMC4515764
- DOI: 10.1038/srep12562
A novel laminin β gene BmLanB1-w regulates wing-specific cell adhesion in silkworm, Bombyx mori
Abstract
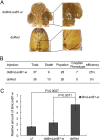
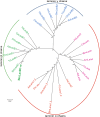
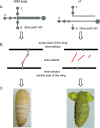
Laminins are important basement membrane (BM) components with crucial roles in development. The numbers of laminin isoforms in various organisms are determined by the composition of the different α, β, and γ chains, and their coding genes, which are variable across spieces. In insects, only two α, one β, and one γ chains have been identified thus far. Here, we isolated a novel laminin β gene, BmLanB1-w, by positional cloning of the mutant (crayfish, cf) with blistered wings in silkworm. Gene structure analysis showed that a 2 bp deletion of the BmLanB1-w gene in the cf mutant caused a frame-shift in the open reading frame (ORF) and generated a premature stop codon. Knockdown of the BmLanB1-w gene produced individuals exhibiting blistered wings, indicating that this laminin gene was required for cell adhesion during wing development. We also identified laminin homologs in different species and showed that two copies of β laminin likely originated in Lepidoptera during evolution. Furthermore, phylogenetic and gene expression analyses of silkworm laminin genes revealed that the BmLanB1-w gene is newly evolved, and is required for wing-specific cell adhesion. This is the first report showing the tissue specific distribution and functional differentiation of β laminin in insects.
Figures




Similar articles
-
A palmitoyltransferase Approximated gene Bm-app regulates wing development in Bombyx mori.Insect Sci. 2020 Feb;27(1):2-13. doi: 10.1111/1744-7917.12629. Epub 2018 Aug 23. Insect Sci. 2020. PMID: 29943911 Free PMC article.
-
BmBlimp-1 gene encoding a C2H2 zinc finger protein is required for wing development in the silkworm Bombyx mori.Int J Biol Sci. 2019 Oct 12;15(12):2664-2675. doi: 10.7150/ijbs.34743. eCollection 2019. Int J Biol Sci. 2019. PMID: 31754338 Free PMC article.
-
BmSd gene regulates the silkworm wing size by affecting the Hippo pathway.Insect Sci. 2020 Aug;27(4):655-664. doi: 10.1111/1744-7917.12702. Epub 2019 Jul 21. Insect Sci. 2020. PMID: 31225693
-
Functional sites in the laminin alpha chains.Connect Tissue Res. 2005;46(3):142-52. doi: 10.1080/03008200591008527. Connect Tissue Res. 2005. PMID: 16147852 Review.
-
Evolution: have wings come, gone and come again?Curr Biol. 2003 May 27;13(11):R436-8. doi: 10.1016/s0960-9822(03)00364-6. Curr Biol. 2003. PMID: 12781152 Review.
Cited by
-
A palmitoyltransferase Approximated gene Bm-app regulates wing development in Bombyx mori.Insect Sci. 2020 Feb;27(1):2-13. doi: 10.1111/1744-7917.12629. Epub 2018 Aug 23. Insect Sci. 2020. PMID: 29943911 Free PMC article.
-
BmBlimp-1 gene encoding a C2H2 zinc finger protein is required for wing development in the silkworm Bombyx mori.Int J Biol Sci. 2019 Oct 12;15(12):2664-2675. doi: 10.7150/ijbs.34743. eCollection 2019. Int J Biol Sci. 2019. PMID: 31754338 Free PMC article.
-
Role of microsatellites in genetic analysis of Bombyx mori silkworm: a review.F1000Res. 2019 Aug 13;8:1424. doi: 10.12688/f1000research.20052.1. eCollection 2019. F1000Res. 2019. PMID: 32148760 Free PMC article. Review.
-
Transcriptomic analysis of the testicular fusion in Spodoptera litura.BMC Genomics. 2020 Feb 19;21(1):171. doi: 10.1186/s12864-020-6494-3. BMC Genomics. 2020. PMID: 32075574 Free PMC article.
-
High-resolution silkworm pan-genome provides genetic insights into artificial selection and ecological adaptation.Nat Commun. 2022 Sep 24;13(1):5619. doi: 10.1038/s41467-022-33366-x. Nat Commun. 2022. PMID: 36153338 Free PMC article.
References
-
- Burgeson R. E. et al. A new nomenclature for the laminins. Matrix Biol 14, 209–211 (1994). - PubMed
-
- Timpl R. et al. Laminin—a glycoprotein from basement membranes. J Biol Chem 254, 9933–9937 (1979). - PubMed
-
- Patarroyo M., Tryggvason K. & Virtanen I. Laminin isoforms in tumor invasion, angiogenesis and metastasis. Semin Cancer Biol 12, 197–207 (2002). - PubMed
-
- Adams J. C. & Watt F. M. Regulation of development and differentiation by the extracellular matrix. Development 117, 1183–1198 (1993). - PubMed
-
- Colognato H. & Yurchenco P. D. Form and function: the laminin family of heterotrimers. Dev Dyn 218, 213–234 (2000). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

