αvβ3 Integrins Mediate Flow-Induced NF-κB Activation, Proinflammatory Gene Expression, and Early Atherogenic Inflammation
- PMID: 26212910
- PMCID: PMC4597278
- DOI: 10.1016/j.ajpath.2015.05.013
αvβ3 Integrins Mediate Flow-Induced NF-κB Activation, Proinflammatory Gene Expression, and Early Atherogenic Inflammation
Abstract
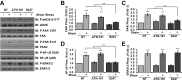
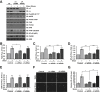
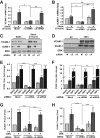
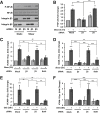
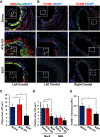
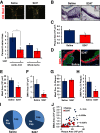
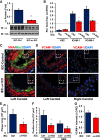
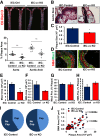
Endothelial cell interactions with transitional matrix proteins, such as fibronectin, occur early during atherogenesis and regulate shear stress-induced endothelial cell activation. Multiple endothelial cell integrins bind transitional matrix proteins, including α5β1, αvβ3, and αvβ5. However, the role these integrins play in mediating shear stress-induced endothelial cell activation remains unclear. Therefore, we sought to elucidate which integrin heterodimers mediate shear stress-induced endothelial cell activation and early atherogenesis. We now show that inhibiting αvβ3 integrins (S247, siRNA), but not α5β1 or αvβ5, blunts shear stress-induced proinflammatory signaling (NF-κB, p21-activated kinase) and gene expression (ICAM1, VCAM1). Importantly, inhibiting αvβ3 did not affect cytokine-induced proinflammatory responses or inhibit all shear stress-induced signaling, because Akt, endothelial nitric oxide synthase, and extracellular regulated kinase activation remained intact. Furthermore, inhibiting αv integrins (S247), but not α5 (ATN-161), in atherosclerosis-prone apolipoprotein E knockout mice significantly reduced vascular remodeling after acute induction of disturbed flow. S247 treatment similarly reduced early diet-induced atherosclerotic plaque formation associated with both diminished inflammation (expression of vascular cell adhesion molecule 1, plaque macrophage content) and reduced smooth muscle incorporation. Inducible, endothelial cell-specific αv integrin deletion similarly blunted inflammation in models of disturbed flow and diet-induced atherogenesis but did not affect smooth muscle incorporation. Our studies identify αvβ3 as the primary integrin heterodimer mediating shear stress-induced proinflammatory responses and as a key contributor to early atherogenic inflammation.
Copyright © 2015 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

