Dexamethasone intravitreal implant in the treatment of diabetic macular edema
- PMID: 26213460
- PMCID: PMC4509543
- DOI: 10.2147/OPTH.S79948
Dexamethasone intravitreal implant in the treatment of diabetic macular edema
Abstract
Diabetic macular edema (DME) resembles a chronic, low-grade inflammatory reaction, and is characterized by blood-retinal barrier (BRB) breakdown and retinal capillary leakage. Corticosteroids are of therapeutic benefit because of their anti-inflammatory, antiangiogenic, and BRB-stabilizing properties. Delivery modes include periocular and intravitreal (via pars plana) injection. To offset the short intravitreal half-life of corticosteroid solutions (~3 hours) and the need for frequent intravitreal injections, sustained-release intravitreal corticosteroid implants have been developed. Dexamethasone intravitreal implant provides retinal drug delivery for ≤6 months and recently has been approved for use in the treatment of DME. Pooled findings (n=1,048) from two large-scale, randomized Phase III trials indicated that dexamethasone intravitreal implant (0.35 mg and 0.7 mg) administered at ≥6-month intervals produced sustained improvements in best-corrected visual acuity (BCVA) and macular edema. Significantly more patients showed a ≥15-letter gain in BCVA at 3 years with dexamethasone intravitreal implant 0.35 mg and 0.7 mg than with sham injection (18.4% and 22.2% vs 12.0%). Anatomical assessments showed rapid and sustained reductions in macular edema and slowing of retinopathy progression. Phase II study findings suggest that dexamethasone intravitreal implant is effective in focal, cystoid, and diffuse DME, in vitrectomized eyes, and in combination with laser therapy. Ocular complications of dexamethasone intravitreal implant in Phase III trials included cataract-related events (66.0% in phakic patients), intraocular pressure elevation ≥25 mmHg (29.7%), conjunctival hemorrhage (23.5%), vitreous hemorrhage (10.0%), macular fibrosis (8.3%), conjunctival hyperemia (7.2%), eye pain (6.1%), vitreous detachment (5.8%), and dry eye (5.8%); injection-related complications (eg, retinal tear/detachment, vitreous loss, endophthalmitis) were infrequent (<2%). Dexamethasone intravitreal implant offers a viable treatment option for DME, especially in cases that are persistent or treatment (anti-vascular endothelial growth factor/laser) refractory.
Keywords: corticosteroids; dexamethasone; diabetic retinopathy; implant; intravitreal; macular edema.
Figures
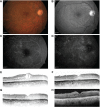
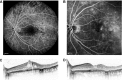
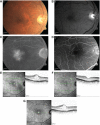

Similar articles
-
Use of intravitreal dexamethasone implant in patients with cataract and macular edema undergoing phacoemulsification.Eur J Ophthalmol. 2015 Mar-Apr;25(2):168-72. doi: 10.5301/ejo.5000523. Epub 2014 Oct 21. Eur J Ophthalmol. 2015. PMID: 25363857
-
Anatomical effects of dexamethasone intravitreal implant in diabetic macular oedema: a pooled analysis of 3-year phase III trials.Br J Ophthalmol. 2016 Jun;100(6):796-801. doi: 10.1136/bjophthalmol-2015-306823. Epub 2015 Nov 18. Br J Ophthalmol. 2016. PMID: 26581718 Free PMC article. Clinical Trial.
-
Dexamethasone intravitreal implant in combination with laser photocoagulation for the treatment of diffuse diabetic macular edema.Ophthalmology. 2013 Sep;120(9):1843-51. doi: 10.1016/j.ophtha.2013.02.018. Epub 2013 May 22. Ophthalmology. 2013. PMID: 23706947 Clinical Trial.
-
Clinical trials on corticosteroids for diabetic macular edema.World J Diabetes. 2013 Dec 15;4(6):295-302. doi: 10.4239/wjd.v4.i6.295. World J Diabetes. 2013. PMID: 24379920 Free PMC article. Review.
-
Corticosteroids for Diabetic Macular Edema.Taiwan J Ophthalmol. 2019 Dec 13;9(4):233-242. doi: 10.4103/tjo.tjo_68_19. eCollection 2019 Oct-Dec. Taiwan J Ophthalmol. 2019. PMID: 31942428 Free PMC article. Review.
Cited by
-
Contralateral eye-to-eye comparison of intravitreal ranibizumab and a sustained-release dexamethasone intravitreal implant in recalcitrant diabetic macular edema.Clin Ophthalmol. 2016 Aug 29;10:1679-84. doi: 10.2147/OPTH.S110789. eCollection 2016. Clin Ophthalmol. 2016. PMID: 27621587 Free PMC article.
-
Intravitreal DEX Implant for the Treatment of Diabetic Macular Edema: A Review of National Consensus.Pharmaceutics. 2023 Oct 13;15(10):2461. doi: 10.3390/pharmaceutics15102461. Pharmaceutics. 2023. PMID: 37896220 Free PMC article. Review.
-
Real-Life Management of Diabetic Macular Edema with Dexamethasone Intravitreal Implant: A Retrospective Analysis of Long-Term Clinical Outcomes.J Ophthalmol. 2020 Apr 10;2020:4860743. doi: 10.1155/2020/4860743. eCollection 2020. J Ophthalmol. 2020. PMID: 32351722 Free PMC article.
-
Intravitreal Dexamethasone Implant as a Sustained Release Drug Delivery Device for the Treatment of Ocular Diseases: A Comprehensive Review of the Literature.Pharmaceutics. 2020 Jul 26;12(8):703. doi: 10.3390/pharmaceutics12080703. Pharmaceutics. 2020. PMID: 32722556 Free PMC article. Review.
-
Inflammation: The Link between Neural and Vascular Impairment in the Diabetic Retina and Therapeutic Implications.Int J Mol Sci. 2023 May 15;24(10):8796. doi: 10.3390/ijms24108796. Int J Mol Sci. 2023. PMID: 37240138 Free PMC article. Review.
References
-
- Klein R, Klein BE, Moss SE, Davis MD, DeMets DL. The Wisconsin epidemiologic study of diabetic retinopathy. IV. Diabetic macular edema. Ophthalmology. 1984;91(12):1464–1474. - PubMed
-
- Antcliff RJ, Marshall J. The pathogenesis of edema in diabetic maculopathy. Semin Ophthalmol. 1999;14(4):223–232. - PubMed
-
- Bhagat N, Grigorian RA, Tutela A, Zarbin MA. Diabetic macular edema: pathogenesis and treatment. Surv Ophthalmol. 2009;54(1):1–32. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

