Pharmacology and physiology of gastrointestinal enteroendocrine cells
- PMID: 26213627
- PMCID: PMC4506687
- DOI: 10.1002/prp2.155
Pharmacology and physiology of gastrointestinal enteroendocrine cells
Abstract
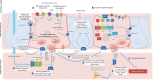
Gastrointestinal (GI) polypeptides are secreted from enteroendocrine cells (EECs). Recent technical advances and the identification of endogenous and synthetic ligands have enabled exploration of the pharmacology and physiology of EECs. Enteroendocrine signaling pathways stimulating hormone secretion involve multiple nutrient transporters and G protein-coupled receptors (GPCRs), which are activated simultaneously under prevailing nutrient conditions in the intestine following a meal. The majority of studies investigate hormone secretion from EECs in response to single ligands and although the mechanisms behind how individual signaling pathways generate a hormonal output have been well characterized, our understanding of how these signaling pathways converge to generate a single hormone secretory response is still in its infancy. However, a picture is beginning to emerge of how nutrients and full, partial, or allosteric GPCR ligands differentially regulate the enteroendocrine system and its interaction with the enteric and central nervous system. So far, activation of multiple pathways underlies drug discovery efforts to harness the therapeutic potential of the enteroendocrine system to mimic the phenotypic changes observed in patients who have undergone Roux-en-Y gastric surgery. Typically obese patients exhibit ∼30% weight loss and greater than 80% of obese diabetics show remission of diabetes. Targeting combinations of enteroendocrine signaling pathways that work synergistically may manifest with significant, differentiated EEC secretory efficacy. Furthermore, allosteric modulators with their increased selectivity, self-limiting activity, and structural novelty may translate into more promising enteroendocrine drugs. Together with the potential to bias enteroendocrine GPCR signaling and/or to activate multiple divergent signaling pathways highlights the considerable range of therapeutic possibilities available. Here, we review the pharmacology and physiology of the EEC system.
Keywords: Chemosensing; GLP-1; GPCR; diabetes; enteroendocrine; intestine.
Figures


References
-
- Adrian TE, Gariballa S, Parekh KA, Thomas SA, Saadi H, Al KJ. Rectal taurocholate increases L cell and insulin secretion, and decreases blood glucose and food intake in obese type 2 diabetic volunteers. Diabetologia. 2012;55:2343–2347. , et al. ( - PubMed
-
- Akhtar M, Watson JL, Nazli A, McKay DM. Bacterial DNA evokes epithelial IL-8 production by a MAPK-dependent, NF-kappaB-independent pathway. FASEB J. 2003;17:1319–1321. - PubMed
-
- Amato A, Cinci L, Rotondo A, Serio R, Faussone-Pellegrini MS, Vannucchi MG. Peripheral motor action of glucagon-like peptide-1 through enteric neuronal receptors. Neurogastroenterol Motil. 2010;22:664-e203. , et al. ( - PubMed
-
- Anini Y, Brubaker PL. Muscarinic receptors control glucagon-like peptide 1 secretion by human endocrine L cells. Endocrinology. 2003;144:3244–3250. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

