One-step biosynthesis of α-ketoisocaproate from L-leucine by an Escherichia coli whole-cell biocatalyst expressing an L-amino acid deaminase from Proteus vulgaris
- PMID: 26217895
- PMCID: PMC4517468
- DOI: 10.1038/srep12614
One-step biosynthesis of α-ketoisocaproate from L-leucine by an Escherichia coli whole-cell biocatalyst expressing an L-amino acid deaminase from Proteus vulgaris
Abstract
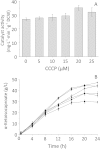
This work aimed to develop a whole-cell biotransformation process for the production of α-ketoisocaproate from L-leucine. A recombinant Escherichia coli strain was constructed by expressing an L-amino acid deaminase from Proteus vulgaris. To enhance α-ketoisocaproate production, the reaction conditions were optimized as follows: whole-cell biocatalyst 0.8 g/L, leucine concentration 13.1 g/L, temperature 35 °C, pH 7.5, and reaction time 20 h. Under the above conditions, the α-ketoisocaproate titer reached 12.7 g/L with a leucine conversion rate of 97.8%. In addition, different leucine feeding strategies were examined to increase the α-ketoisocaproate titer. When 13.1 g/L leucine was added at 2-h intervals (from 0 to 22 h, 12 addition times), the α-ketoisocaproate titer reached 69.1 g/L, while the leucine conversion rate decreased to 50.3%. We have developed an effective process for the biotechnological production of α-ketoisocaproate that is more environmentally friendly than the traditional petrochemical synthesis approach.
Figures

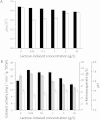
 : 0 μM,
: 0 μM,  : 5 μM,
: 5 μM,  : 10 μM,
: 10 μM,  : 15 μM,
: 15 μM,  : 20 μM,
: 20 μM,  : 25 μM.
: 25 μM.
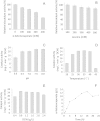
 : 2 h,
: 2 h,  : 4 h,
: 4 h,  : 6 h,
: 6 h,  : 8 h;
: 8 h;  : 10 h.
: 10 h.
 ) for free cell and dot line with triangle mark (
) for free cell and dot line with triangle mark ( ) for immobilized cell]; Comparison of the pH (C), and thermal (D) (black bar for free and light grey bar for immobilized recombinant whole cell biocatalyst).
) for immobilized cell]; Comparison of the pH (C), and thermal (D) (black bar for free and light grey bar for immobilized recombinant whole cell biocatalyst).Similar articles
-
One-step biosynthesis of α-keto-γ-methylthiobutyric acid from L-methionine by an Escherichia coli whole-cell biocatalyst expressing an engineered L-amino acid deaminase from Proteus vulgaris.PLoS One. 2014 Dec 22;9(12):e114291. doi: 10.1371/journal.pone.0114291. eCollection 2014. PLoS One. 2014. PMID: 25531756 Free PMC article.
-
Tuning the transcription and translation of L-amino acid deaminase in Escherichia coli improves α-ketoisocaproate production from L-leucine.PLoS One. 2017 Jun 29;12(6):e0179229. doi: 10.1371/journal.pone.0179229. eCollection 2017. PLoS One. 2017. PMID: 28662040 Free PMC article.
-
Production of α-ketoisocaproate via free-whole-cell biotransformation by Rhodococcus opacus DSM 43250 with L-leucine as the substrate.Enzyme Microb Technol. 2011 Sep 10;49(4):321-5. doi: 10.1016/j.enzmictec.2011.06.008. Epub 2011 Jun 15. Enzyme Microb Technol. 2011. PMID: 22112557
-
Engineering of L-amino acid deaminases for the production of α-keto acids from L-amino acids.Bioengineered. 2019 Dec;10(1):43-51. doi: 10.1080/21655979.2019.1595990. Bioengineered. 2019. PMID: 30876377 Free PMC article. Review.
-
Biotechnological production of alpha-keto acids: Current status and perspectives.Bioresour Technol. 2016 Nov;219:716-724. doi: 10.1016/j.biortech.2016.08.015. Epub 2016 Aug 8. Bioresour Technol. 2016. PMID: 27575335 Review.
Cited by
-
Dual contribution of the mTOR pathway and of the metabolism of amino acids in prostate cancer.Cell Oncol (Dordr). 2022 Oct;45(5):831-859. doi: 10.1007/s13402-022-00706-4. Epub 2022 Aug 29. Cell Oncol (Dordr). 2022. PMID: 36036882 Review.
-
Highly selective synthesis of d-amino acids from readily available l-amino acids by a one-pot biocatalytic stereoinversion cascade.RSC Adv. 2019 Sep 23;9(51):29927-29935. doi: 10.1039/c9ra06301c. eCollection 2019 Sep 18. RSC Adv. 2019. PMID: 35531513 Free PMC article.
-
Production of Salvianic Acid A from l-DOPA via Biocatalytic Cascade Reactions.Molecules. 2022 Sep 18;27(18):6088. doi: 10.3390/molecules27186088. Molecules. 2022. PMID: 36144828 Free PMC article.
-
Semi-rational engineering membrane binding domain of L-amino acid deaminase from Proteus vulgaris for enhanced α-ketoisocaproate.Front Microbiol. 2022 Sep 30;13:1025845. doi: 10.3389/fmicb.2022.1025845. eCollection 2022. Front Microbiol. 2022. PMID: 36246292 Free PMC article.
-
Membrane binding of the insertion sequence of Proteus vulgaris L-amino acid deaminase stabilizes protein structure and increases catalytic activity.Sci Rep. 2017 Oct 20;7(1):13719. doi: 10.1038/s41598-017-14238-7. Sci Rep. 2017. PMID: 29057984 Free PMC article.
References
-
- Aparicio M. et al. Keto acid therapy in predialysis chronic kidney disease patients: final consensus. J. Renal. Nutr. 22, S22–S24 (2012). - PubMed
-
- Aparicio M. et al. Protein-restricted diets plus keto/amino acids-a valid therapeutic approach for chronic kidney disease patients. J. Renal. Nutr. 22, S1–S21 (2012). - PubMed
-
- Mou S., Li J., Yu Z., Wang Q. & Ni Z. Keto acid-supplemented low-protein diet for treatment of adult patients with hepatitis B virus infection and chronic glomerulonephritis. J. Int. Med. Res. 41, 129–137 (2013). - PubMed
-
- Holecek M. Three targets of branched-chain amino acid supplementation in the treatment of liver disease. Nutrition. 26, 482–490 (2010). - PubMed
-
- Chua B., Siehl D. L. & Morgan H. E. Effect of leucine and metabolites of branched chain amino acids on protein turnover in heart. J. Biol. Chem. 254, 8358–8362 (1979). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

