Idiopathic preretinal glia in aging and age-related macular degeneration
- PMID: 26220834
- PMCID: PMC4728061
- DOI: 10.1016/j.exer.2015.07.016
Idiopathic preretinal glia in aging and age-related macular degeneration
Abstract
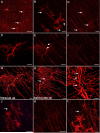
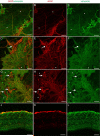
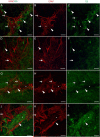
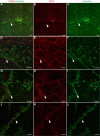
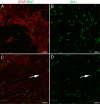
During analysis of glia in wholemount aged human retinas, frequent projections onto the vitreal surface of the inner limiting membrane (ILM) were noted. The present study characterized these preretinal glial structures. The amount of glial cells on the vitreal side of the ILM was compared between eyes with age-related macular degeneration (AMD) and age-matched control eyes. Retinal wholemounts were stained for markers of retinal astrocytes and activated Müller cells (glial fibrillary acidic protein, GFAP), Müller cells (vimentin, glutamine synthetase) and microglia/hyalocytes (IBA-1). Retinal vessels were labeled with UEA lectin. Images were collected using a Zeiss LSM 710 confocal microscope. Retinas were then cryopreserved. Laminin labeling of cryosections determined the location of glial structures in relation to the ILM. All retinas investigated herein had varied amounts of preretinal glia. These glial structures were classified into three groups based on size: sprouts, blooms, and membranes. The simplest of the glial structures observed were focal sprouts of singular GFAP-positive cells or processes on the vitreal surface of the ILM. The intermediate structures observed, glial blooms, were created by multiple cells/processes exiting from a single point and extending along the vitreoretinal surface. The most extensive structures, glial membranes, consisted of compact networks of cells and processes. Preretinal glia were observed in all areas of the retina but they were most prominent over large vessels. While all glial blooms and membranes contained vimentin and GFAP-positive cells, these proteins did not always co-localize. Many areas had no preretinal GFAP but had numerous vimentin only glial sprouts. In double labeled glial sprouts, vimentin staining extended beyond that of GFAP. Hyalocytes and microglia were detected along with glial sprouts, blooms, and membranes. They did not, however, concentrate in the retina below these structures. Cross sectional analysis identified small breaks in the ILM above large retinal vessels through which glial cells exited the retina. Preretinal glial structures of varied sizes are a common occurrence in aged retinas and, in most cases, are subclinical. While all retinal glia are found in blooms, vimentin labeling suggests that Müller cells form the leading edge. All retinas investigated from eyes with active choroidal neovascularization (CNV) had extensive glial membranes on the vitreal surface of the ILM. Although these structures may be benign, they may exert traction on the retina as they spread along the vitreoretinal interface. In cases with CNV, glial cells in the vitreous could bind intravitreally injected anti-vascular endothelial growth factor. These preretinal glial structures indicate the remodeling of both astrocytes and Müller cells in aged retinas, in particular those with advanced AMD.
Keywords: Age-related macular degeneration; Astrocytes; Epiretinal membranes; Glia; Muller cells.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures






References
-
- Blasiak J, Salminen A, Kaarniranta K. Potential of epigenetic mechanisms in AMD pathology. Front Biosci (Schol Ed) 2013;5:412–425. - PubMed
-
- Bringmann A, Wiedemann P. Involvement of Muller glial cells in epiretinal membrane formation. Graefes Arch Clin Exp Ophthalmol. 2009;247:865–883. - PubMed
-
- Charteris DG, Sethi CS, Lewis GP, Fisher SK. Proliferative vitreoretinopathy-developments in adjunctive treatment and retinal pathology. Eye. 2002;16:369–374. - PubMed
-
- Fisher SK, Lewis GP. Muller cell and neuronal remodeling in retinal detachment and reattachment and their potential consequences for visual recovery: a review and reconsideration of recent data. Vision Res. 2003;43:887–897. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

