Long-Term Effects of Low-Dose Spironolactone on Chronic Dialysis Patients: A Randomized Placebo-Controlled Study
- PMID: 26224543
- PMCID: PMC8031645
- DOI: 10.1111/jch.12628
Long-Term Effects of Low-Dose Spironolactone on Chronic Dialysis Patients: A Randomized Placebo-Controlled Study
Abstract
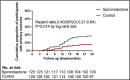
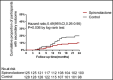
The purpose of this 2-year multicentric, randomized, placebo-controlled study was to evaluate the long-term effects and adverse effects of spironolactone on chronic dialysis patients. A total of 253 non-heart failure dialysis patients with end-stage renal disease were randomly assigned to 2-year treatment with spironolactone (25 mg once daily, n=125) or a matching placebo (n=128) as add-on therapy. The primary outcome was a composite of death from cardiocerebrovascular (CCV) events, aborted cardiac arrest, and sudden cardiac death, and the secondary outcome was death from all causes. Other CCV-related indexes such as left ventricular mass index, left ventricular ejection fraction, heart rate variability, vascular endothelial function, and blood pressure-lowering effect were analyzed for patients who completed the whole 2-year follow-up study. Sociodemographic, clinical, and relevant laboratory data were also collected. During the 2-year follow-up, the primary outcome occurred less frequently in the spironolactone group vs the control group (7.2% vs 18.0%; adjusted hazard ratio [HR], 0.42; 95% confidence interval [CI], 0.26-0.78). Death from CCV events occurred in 4.0% of patients in the spironolactone group and in 11.7% of patients in the control group. Neither aborted cardiac arrest nor sudden cardiac death was significantly reduced by spironolactone treatment. The secondary outcome occurred less frequently in the spironolactone group vs the control group (9.6% vs 19.5%; adjusted HR, 0.52; 95% CI, 0.29-0.94). Other CCV-related indexes except for heart rate variability were significantly improved. This study demonstrates that use of low-dose spironolactone in non-heart failure dialysis patients can effectively reduce the risks of both CCV morbidity and mortality with few side effects. Moreover, the beneficial effect was mediated through improving the endothelial function or reducing left ventricular size independent of blood pressure changes, rather than mediation through changes in salt or potassium handling in the kidney.
©2015 Wiley Periodicals, Inc.
Figures
Comment in
-
Mineralocorticoid Receptor Antagonism for Cardiovascular Protection in End-Stage Renal Disease: New Data But the Controversy Continues.J Clin Hypertens (Greenwich). 2016 Mar;18(3):197-9. doi: 10.1111/jch.12718. Epub 2015 Oct 12. J Clin Hypertens (Greenwich). 2016. PMID: 26456776 Free PMC article. No abstract available.
References
-
- Toyoda K, Ninomiya T. Stroke and cerebrovascular diseases in patients with chronic kidney disease. Lancet Neurol. 2014;13:823–833. - PubMed
-
- Power A. Stroke in dialysis and chronic kidney disease. Blood Purif. 2013;36:179–183. - PubMed
-
- Noris M, Remuzzi G. Cardiovascular complications in atypical haemolytic uraemic syndrome. Nat Rev Nephrol. 2014;10:174–180. - PubMed
-
- Matsumoto Y, Mori Y, Kageyama S, et al. Spironolactone reduces cardiovascular and cerebrovascular morbidity and mortality in hemodialysis patients. J Am Coll Cardiol. 2014;63:528–536. - PubMed
-
- Pelliccia F, Rosano G, Patti G, et al. Efficacy and safety of mineralocorticoid receptors in mild to moderate arterial hypertension. Int J Cardiol 2015;200:8–11. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical