Losing Control: Excessive Alcohol Seeking after Selective Inactivation of Cue-Responsive Neurons in the Infralimbic Cortex
- PMID: 26224858
- PMCID: PMC6605108
- DOI: 10.1523/JNEUROSCI.0684-15.2015
Losing Control: Excessive Alcohol Seeking after Selective Inactivation of Cue-Responsive Neurons in the Infralimbic Cortex
Abstract
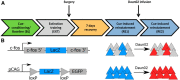
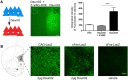
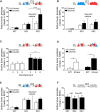
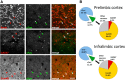
Loss of control over drinking is a key deficit in alcoholism causally associated with malfunction of the medial prefrontal cortex (mPFC), but underlying molecular and cellular mechanisms remain unclear. Cue-induced reinstatement of alcohol seeking activates a subset of mPFC neurons in rats, identified by their common expression of the activity marker cFos and comprised of both principal and interneurons. Here, we used cFos-lacZ and pCAG-lacZ transgenic rats for activity-dependent or nonselective inactivation of neurons, respectively, which by their lacZ encoded β-galactosidase activity convert the inactive prodrug Daun02 into the neurotoxin daunorubicin. We report that activity-dependent ablation of a neuronal ensemble in the infralimbic but not the prelimbic subregion induced excessive alcohol seeking. The targeted neuronal ensemble was specific for the cue-induced response because stress-induced reinstatement was not affected in these animals. Importantly, nonselective inactivation of infralimbic neurons, using pCAG-lacZ rats, was without functional consequence on the cue-induced reinstatement task. Thus, inhibitory control over alcohol seeking is exerted by distinct functional ensembles within the infralimbic cortex rather than by a general inhibitory tone of this region on the behavioral output. This indicates a high level of functional compartmentation within the rat mPFC whereat many functional ensembles could coexist and interact within the same subregion.
Significance statement: Hebb's (1949) idea of memories as being represented in local neuronal networks is supported by identification of transiently stable activity patterns within subgroups of neurons. However, it is difficult to link individual networks to specific memory tasks, for example a learned behavior. By a novel approach of activity-dependent ablation, here we identify a specific neuronal ensemble located in the infralimbic subregion of the medial prefrontal cortex that controls a seeking response for alcohol in rats. Our data demonstrate that functional output depends on specific neuronal ensembles within a given brain region rather than on the global activity of that region, which raises important questions about the interpretation of numerous earlier experiments using site-directed silencing or stimulation for elucidating brain function.
Keywords: Daun02 inactivation method; alcohol self-administration; conditioned cues; medial prefrontal cortex; neuronal ensembles; reinstatement.
Copyright © 2015 the authors 0270-6474/15/3510750-12$15.00/0.
Figures


Similar articles
-
Choice for Drug or Natural Reward Engages Largely Overlapping Neuronal Ensembles in the Infralimbic Prefrontal Cortex.J Neurosci. 2018 Apr 4;38(14):3507-3519. doi: 10.1523/JNEUROSCI.0026-18.2018. Epub 2018 Feb 26. J Neurosci. 2018. PMID: 29483279 Free PMC article.
-
Separate vmPFC Ensembles Control Cocaine Self-Administration Versus Extinction in Rats.J Neurosci. 2019 Sep 11;39(37):7394-7407. doi: 10.1523/JNEUROSCI.0918-19.2019. Epub 2019 Jul 22. J Neurosci. 2019. PMID: 31331999 Free PMC article.
-
Regulation of Alcohol Extinction and Cue-Induced Reinstatement by Specific Projections among Medial Prefrontal Cortex, Nucleus Accumbens, and Basolateral Amygdala.J Neurosci. 2017 Apr 26;37(17):4462-4471. doi: 10.1523/JNEUROSCI.3383-16.2017. Epub 2017 Mar 23. J Neurosci. 2017. PMID: 28336571 Free PMC article.
-
Cortical and amygdalar neuronal ensembles in alcohol seeking, drinking and withdrawal.Neuropharmacology. 2017 Aug 1;122:107-114. doi: 10.1016/j.neuropharm.2017.04.031. Epub 2017 Apr 20. Neuropharmacology. 2017. PMID: 28435008 Free PMC article. Review.
-
Learning-induced intrinsic and synaptic plasticity in the rodent medial prefrontal cortex.Neurobiol Learn Mem. 2020 Mar;169:107117. doi: 10.1016/j.nlm.2019.107117. Epub 2019 Nov 23. Neurobiol Learn Mem. 2020. PMID: 31765801 Free PMC article. Review.
Cited by
-
Fos-expressing neuronal ensemble in rat ventromedial prefrontal cortex encodes cocaine seeking but not food seeking in rats.Addict Biol. 2021 May;26(3):e12943. doi: 10.1111/adb.12943. Epub 2020 Jul 19. Addict Biol. 2021. PMID: 32683756 Free PMC article.
-
Neural circuits controlling choice behavior in opioid addiction.Neuropharmacology. 2023 Mar 15;226:109407. doi: 10.1016/j.neuropharm.2022.109407. Epub 2022 Dec 30. Neuropharmacology. 2023. PMID: 36592884 Free PMC article. Review.
-
Chemogenetic Tools for Causal Cellular and Neuronal Biology.Physiol Rev. 2018 Jan 1;98(1):391-418. doi: 10.1152/physrev.00009.2017. Physiol Rev. 2018. PMID: 29351511 Free PMC article. Review.
-
Approaches and considerations of studying neuronal ensembles: a brief review.Front Cell Neurosci. 2023 Dec 14;17:1310724. doi: 10.3389/fncel.2023.1310724. eCollection 2023. Front Cell Neurosci. 2023. PMID: 38155864 Free PMC article. Review.
-
Distinct memory engrams in the infralimbic cortex of rats control opposing environmental actions on a learned behavior.Elife. 2016 Dec 10;5:e21920. doi: 10.7554/eLife.21920. Elife. 2016. PMID: 27938664 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
