Grading of Gliomas by Using Monoexponential, Biexponential, and Stretched Exponential Diffusion-weighted MR Imaging and Diffusion Kurtosis MR Imaging
- PMID: 26230975
- PMCID: PMC4734159
- DOI: 10.1148/radiol.2015142173
Grading of Gliomas by Using Monoexponential, Biexponential, and Stretched Exponential Diffusion-weighted MR Imaging and Diffusion Kurtosis MR Imaging
Abstract
Purpose: To quantitatively compare the potential of various diffusion parameters obtained from monoexponential, biexponential, and stretched exponential diffusion-weighted imaging models and diffusion kurtosis imaging in the grading of gliomas.
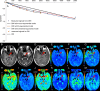
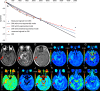
Materials and methods: This study was approved by the local ethics committee, and written informed consent was obtained from all subjects. Both diffusion-weighted imaging and diffusion kurtosis imaging were performed in 69 patients with pathologically proven gliomas by using a 3-T magnetic resonance (MR) imaging unit. An isotropic apparent diffusion coefficient (ADC), true ADC, pseudo-ADC, and perfusion fraction were calculated from diffusion-weighted images by using a biexponential model. A water molecular diffusion heterogeneity index and distributed diffusion coefficient were calculated from diffusion-weighted images by using a stretched exponential model. Mean diffusivity, fractional anisotropy, and mean kurtosis were calculated from diffusion kurtosis images. All values were compared between high-grade and low-grade gliomas by using a Mann-Whitney U test. Receiver operating characteristic and Spearman rank correlation analysis were used for statistical evaluations.
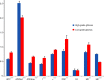
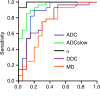
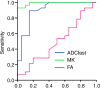
Results: ADC, true ADC, perfusion fraction, water molecular diffusion heterogeneity index, distributed diffusion coefficient, and mean diffusivity values were significantly lower in high-grade gliomas than in low-grade gliomas (U = 109, 56, 129, 6, 206, and 229, respectively; P < .05). Pseudo-ADC and mean kurtosis values were significantly higher in high-grade gliomas than in low-grade gliomas (U = 98 and 8, respectively; P < .05). Both water molecular diffusion heterogeneity index (area under the receiver operating characteristic curve [AUC] = 0.993) and mean kurtosis (AUC = 0.991) had significantly greater AUC values than ADC (AUC = 0.866), mean diffusivity (AUC = 0.722), and fractional anisotropy (AUC = 0.500) in the differentiation of low-grade and high-grade gliomas (P < .05).
Conclusion: Water molecular diffusion heterogeneity index and mean kurtosis values may provide additional information and improve the grading of gliomas compared with conventional diffusion parameters.
© RSNA, 2015.
Figures
Similar articles
-
Grading and proliferation assessment of diffuse astrocytic tumors with monoexponential, biexponential, and stretched-exponential diffusion-weighted imaging and diffusion kurtosis imaging.Eur J Radiol. 2018 Dec;109:188-195. doi: 10.1016/j.ejrad.2018.11.003. Epub 2018 Nov 5. Eur J Radiol. 2018. PMID: 30527302
-
Gliomas: diffusion kurtosis MR imaging in grading.Radiology. 2012 May;263(2):492-501. doi: 10.1148/radiol.12110927. Epub 2012 Mar 8. Radiology. 2012. PMID: 22403168
-
Measurements of diagnostic examination performance using quantitative apparent diffusion coefficient and proton MR spectroscopic imaging in the preoperative evaluation of tumor grade in cerebral gliomas.Eur J Radiol. 2011 Nov;80(2):462-70. doi: 10.1016/j.ejrad.2010.07.017. Epub 2010 Aug 13. Eur J Radiol. 2011. PMID: 20708868
-
Diagnostic Performance of Monoexponential DWI Versus Diffusion Kurtosis Imaging in Prostate Cancer: A Systematic Review and Meta-Analysis.AJR Am J Roentgenol. 2018 Aug;211(2):358-368. doi: 10.2214/AJR.17.18934. Epub 2018 May 29. AJR Am J Roentgenol. 2018. PMID: 29812977
-
The utility of diffusion MRI with quantitative ADC measurements for differentiating high-grade from low-grade cerebral gliomas: Evidence from a meta-analysis.J Neurol Sci. 2017 Feb 15;373:9-15. doi: 10.1016/j.jns.2016.12.008. Epub 2016 Dec 9. J Neurol Sci. 2017. PMID: 28131237 Review.
Cited by
-
Radiogenomics correlation between MR imaging features and mRNA-based subtypes in lower-grade glioma.BMC Neurol. 2020 Jun 29;20(1):259. doi: 10.1186/s12883-020-01838-6. BMC Neurol. 2020. PMID: 32600353 Free PMC article.
-
Heterogeneity Diffusion Imaging of gliomas: Initial experience and validation.PLoS One. 2019 Nov 14;14(11):e0225093. doi: 10.1371/journal.pone.0225093. eCollection 2019. PLoS One. 2019. PMID: 31725772 Free PMC article.
-
Diffusion Kurtosis Imaging as a Tool in Neurotoxicology.Neurotox Res. 2020 Jan;37(1):41-47. doi: 10.1007/s12640-019-00100-3. Epub 2019 Aug 17. Neurotox Res. 2020. PMID: 31422570
-
Design and validation of diffusion MRI models of white matter.Front Phys. 2017 Nov;28:61. doi: 10.3389/fphy.2017.00061. Epub 2017 Nov 28. Front Phys. 2017. PMID: 29755979 Free PMC article.
-
Diffusion and quantification of diffusion of prostate cancer.Br J Radiol. 2022 Mar 1;95(1131):20210653. doi: 10.1259/bjr.20210653. Epub 2021 Sep 19. Br J Radiol. 2022. PMID: 34538094 Free PMC article. Review.
References
-
- Scott JN, Brasher PM, Sevick RJ, Rewcastle NB, Forsyth PA. How often are nonenhancing supratentorial gliomas malignant? A population study. Neurology 2002;59(6):947–949. - PubMed
-
- Sugahara T, Korogi Y, Kochi M, et al. . Usefulness of diffusion-weighted MRI with echo-planar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging 1999;9(1):53–60. - PubMed
-
- Stadlbauer A, Ganslandt O, Buslei R, et al. . Gliomas: histopathologic evaluation of changes in directionality and magnitude of water diffusion at diffusion-tensor MR imaging. Radiology 2006;240(3):803–810. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

