Amblyomma maculatum Feeding Augments Rickettsia parkeri Infection in a Rhesus Macaque Model: A Pilot Study
- PMID: 26244337
- PMCID: PMC4526656
- DOI: 10.1371/journal.pone.0135175
Amblyomma maculatum Feeding Augments Rickettsia parkeri Infection in a Rhesus Macaque Model: A Pilot Study
Erratum in
-
Correction: Amblyomma maculatum Feeding Augments Rickettsia parkeri Infection in a Rhesus Macaque Model: A Pilot Study.PLoS One. 2015 Aug 31;10(8):e0137598. doi: 10.1371/journal.pone.0137598. eCollection 2015. PLoS One. 2015. PMID: 26322516 Free PMC article. No abstract available.
Abstract
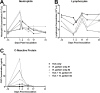
Rickettsia parkeri is an emerging eschar-causing human pathogen in the spotted fever group of Rickettsia and is transmitted by the Gulf coast tick, Amblyomma maculatum. Tick saliva has been shown to alter both the cellular and humoral components of the innate and adaptive immune systems. However, the effect of this immunomodulation on Rickettsia transmission and pathology in an immunocompetent vertebrate host has not been fully examined. We hypothesize that, by modifying the host immune response, tick feeding enhances infection and pathology of pathogenic spotted fever group Rickettsia sp. In order to assess this interaction in vivo, a pilot study was conducted using five rhesus macaques that were divided into three groups. One group was intradermally inoculated with low passage R. parkeri (Portsmouth strain) alone (n = 2) and another group was inoculated during infestation by adult, R. parkeri-free A. maculatum (n = 2). The final macaque was infested with ticks alone (tick feeding control group). Blood, lymph node and skin biopsies were collected at several time points post-inoculation/infestation to assess pathology and quantify rickettsial DNA. As opposed to the tick-only animal, all Rickettsia-inoculated macaques developed inflammatory leukograms, elevated C-reactive protein concentrations, and elevated TH1 (interferon-γ, interleukin-15) and acute phase inflammatory cytokines (interleukin-6) post-inoculation, with greater neutrophilia and interleukin-6 concentrations in the tick plus R. parkeri group. While eschars formed at all R. parkeri inoculation sites, larger and slower healing eschars were observed in the tick feeding plus R. parkeri group. Furthermore, dissemination of R. parkeri to draining lymph nodes early in infection and increased persistence at the inoculation site were observed in the tick plus R. parkeri group. This study indicates that rhesus macaques can be used to model R. parkeri rickettsiosis, and suggests that immunomodulatory factors introduced during tick feeding may enhance the pathogenicity of spotted fever group Rickettsia.
Conflict of interest statement
Figures





References
-
- Adams DA, Jajosky RA, Ajani U, Kriseman J, Sharp P, Onwen DH, et al. Summary of notifiable diseases—United States, 2012. MMWR Morb Mortal Wkly Rep. 2014; 61: 1–121. - PubMed
-
- Paddock CD, Sumner JW, Comer JA, Zaki SR, Goldsmith CS, Goddard J, et al. Rickettsia parkeri: a newly recognized cause of spotted fever rickettsiosis in the United States. Clin Infect Dis. 2004; 38: 805–811. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

