Time-Dependent Effects of Ethanol on BK Channel Expression and Trafficking in Hippocampal Neurons
- PMID: 26247146
- PMCID: PMC4873156
- DOI: 10.1111/acer.12808
Time-Dependent Effects of Ethanol on BK Channel Expression and Trafficking in Hippocampal Neurons
Abstract
Background: The large conductance Ca(2+) - and voltage-activated K(+) channel (BK) is an important player in molecular and behavioral alcohol tolerance. Trafficking and surface expression of ion channels contribute to the development of addictive behaviors. We have previously reported that internalization of the BK channel is a component of molecular tolerance to ethanol (EtOH).
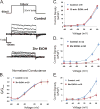
Methods: Using primary cultures of hippocampal neurons, we combine total internal reflection fluorescence microscopy, electrophysiology, and biochemical techniques to explore how exposure to EtOH affects the expression and subcellular localization of endogenous BK channels over time.
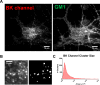
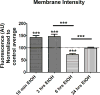
Results: Exposure to EtOH changed the expression of endogenous BK channels in a time-dependent manner at the perimembrane area (plasma membrane and/or the area adjacent to it), while total protein levels of BK remain unchanged. These results suggest a redistribution of the channel within the neurons rather than changes in synthesis or degradation rates. Our results showed a temporally nonlinear effect of EtOH on perimembrane expression of BK. First, there was an increase in BK perimembrane expression after 10 minutes of EtOH exposure that remained evident after 3 hours, although not correlated to increases in functional channel expression. In contrast, after 6 hours of EtOH exposure, we observed a significant decrease in both BK perimembrane expression and functional channel expression. Furthermore, after 24 hours of EtOH exposure, perimembrane levels of BK had returned to baseline.
Conclusions: We report a complex time-dependent pattern in the effect of EtOH on BK channel trafficking, including successive increases and decreases in perimembrane expression and a reduction in active BK channels after 3 and 6 hours of EtOH exposure. Possible mechanisms underlying this multiphasic trafficking are discussed. As molecular tolerance necessarily underlies behavioral tolerance, the time-dependent alterations we see at the level of the channel may be relevant to the influence of drinking patterns on the development of behavioral tolerance.
Keywords: Alcohol; BK Channel; Ethanol; Hippocampus; Tolerance; Trafficking.
Copyright © 2015 by the Research Society on Alcoholism.
Figures







Similar articles
-
Ethanol Effect on BK Channels is Modulated by Magnesium.Alcohol Clin Exp Res. 2015 Sep;39(9):1671-9. doi: 10.1111/acer.12821. Alcohol Clin Exp Res. 2015. PMID: 26331878 Free PMC article.
-
Somatic localization of a specific large-conductance calcium-activated potassium channel subtype controls compartmentalized ethanol sensitivity in the nucleus accumbens.J Neurosci. 2004 Jul 21;24(29):6563-72. doi: 10.1523/JNEUROSCI.0684-04.2004. J Neurosci. 2004. PMID: 15269268 Free PMC article.
-
Alcohol Regulates BK Surface Expression via Wnt/β-Catenin Signaling.J Neurosci. 2016 Oct 12;36(41):10625-10639. doi: 10.1523/JNEUROSCI.0491-16.2016. J Neurosci. 2016. PMID: 27733613 Free PMC article.
-
Sizing up ethanol-induced plasticity: the role of small and large conductance calcium-activated potassium channels.Alcohol Clin Exp Res. 2009 Jul;33(7):1125-35. doi: 10.1111/j.1530-0277.2009.00936.x. Epub 2009 Apr 9. Alcohol Clin Exp Res. 2009. PMID: 19389201 Free PMC article. Review.
-
BK Channels: mediators and models for alcohol tolerance.Trends Neurosci. 2009 Dec;32(12):629-37. doi: 10.1016/j.tins.2009.08.001. Epub 2009 Sep 24. Trends Neurosci. 2009. PMID: 19781792 Free PMC article. Review.
Cited by
-
Forced Abstinence from Volitional Ethanol Intake Drives a Vulnerable Period of Hyperexcitability in BNST-Projecting Insular Cortex Neurons.J Neurosci. 2024 Jan 24;44(4):e1121232023. doi: 10.1523/JNEUROSCI.1121-23.2023. J Neurosci. 2024. PMID: 38050120 Free PMC article.
-
Ethanol's interaction with BK channel α subunit residue K361 does not mediate behavioral responses to alcohol in mice.Mol Psychiatry. 2024 Feb;29(2):529-542. doi: 10.1038/s41380-023-02346-y. Epub 2023 Dec 22. Mol Psychiatry. 2024. PMID: 38135755 Free PMC article.
-
BK channels and alcohol tolerance: Insights from studies on Drosophila, nematodes, rodents and cell lines: A systematic review.Med Int (Lond). 2025 Apr 2;5(4):33. doi: 10.3892/mi.2025.232. eCollection 2025 Jul-Aug. Med Int (Lond). 2025. PMID: 40236633 Free PMC article.
-
Modulation of BK Channels by Ethanol.Int Rev Neurobiol. 2016;128:239-79. doi: 10.1016/bs.irn.2016.03.019. Epub 2016 May 12. Int Rev Neurobiol. 2016. PMID: 27238266 Free PMC article. Review.
-
Large conductance voltage-and calcium-activated K+ (BK) channel in health and disease.Front Pharmacol. 2024 Mar 22;15:1373507. doi: 10.3389/fphar.2024.1373507. eCollection 2024. Front Pharmacol. 2024. PMID: 38584598 Free PMC article. Review.
References
-
- Ahmed SH, Koob GF. Transition from moderate to excessive drug intake: change in hedonic set point. Science (New York, NY) 1998;282(5387):298–300. - PubMed
-
- Carlson SL, Kumar S, Werner DF, Comerford CE, Morrow AL. Ethanol activation of protein kinase A regulates GABAA α1 receptor function and trafficking in cultured cerebral cortical neurons. The Journal of Pharmacology and Experimental Therapeutics. 2013;345(2):317–25. doi: 10.1124/jpet.112.201954. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
- 8G12MD007600/MD/NIMHD NIH HHS/United States
- DA023444/DA/NIDA NIH HHS/United States
- R25 GM061838/GM/NIGMS NIH HHS/United States
- R01 AA017920/AA/NIAAA NIH HHS/United States
- R01 DA037924/DA/NIDA NIH HHS/United States
- DA037924/DA/NIDA NIH HHS/United States
- K99 DA023444/DA/NIDA NIH HHS/United States
- R00 DA023444/DA/NIDA NIH HHS/United States
- P20 GM103642/GM/NIGMS NIH HHS/United States
- 5P20GM103642/GM/NIGMS NIH HHS/United States
- G12 MD007600/MD/NIMHD NIH HHS/United States
- 1R01 AA017920/AA/NIAAA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

