Conformational Motions and Functionally Key Residues for Vitamin B12 Transporter BtuCD-BtuF Revealed by Elastic Network Model with a Function-Related Internal Coordinate
- PMID: 26247943
- PMCID: PMC4581229
- DOI: 10.3390/ijms160817933
Conformational Motions and Functionally Key Residues for Vitamin B12 Transporter BtuCD-BtuF Revealed by Elastic Network Model with a Function-Related Internal Coordinate
Abstract
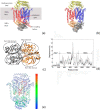
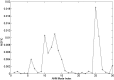
BtuCD-BtuF from Escherichia coli is a binding protein-dependent adenosine triphosphate (ATP)-binding cassette (ABC) transporter system that uses the energy of ATP hydrolysis to transmit vitamin B12 across cellular membranes. Experimental studies have showed that during the transport cycle, the transporter undergoes conformational transitions between the "inward-facing" and "outward-facing" states, which results in the open-closed motions of the cytoplasmic gate of the transport channel. The opening-closing of the channel gate play critical roles for the function of the transporter, which enables the substrate vitamin B12 to be translocated into the cell. In the present work, the extent of opening of the cytoplasmic gate was chosen as a function-related internal coordinate. Then the mean-square fluctuation of the internal coordinate, as well as the cross-correlation between the displacement of the internal coordinate and the movement of each residue in the protein, were calculated based on the normal mode analysis of the elastic network model to analyze the function-related motions encoded in the structure of the system. In addition, the key residues important for the functional motions of the transporter were predicted by using a perturbation method. In order to facilitate the calculations, the internal coordinate was introduced as one of the axes of the coordinate space and the conventional Cartesian coordinate space was transformed into the internal/Cartesian space with linear approximation. All the calculations were carried out in this internal/Cartesian space. Our method can successfully identify the functional motions and key residues for the transporter BtuCD-BtuF, which are well consistent with the experimental observations.
Keywords: ABC transporter; BtuCD–BtuF; elastic network model; functional motions; internal coordinate; key residues; normal mode analysis; perturbation method.
Figures


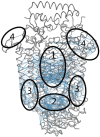
Similar articles
-
Holo-BtuF stabilizes the open conformation of the vitamin B12 ABC transporter BtuCD.Proteins. 2010 Feb 15;78(3):738-53. doi: 10.1002/prot.22606. Proteins. 2010. PMID: 19847921
-
Asymmetry in the structure of the ABC transporter-binding protein complex BtuCD-BtuF.Science. 2007 Sep 7;317(5843):1387-90. doi: 10.1126/science.1145950. Epub 2007 Aug 2. Science. 2007. PMID: 17673622
-
In vitro functional characterization of BtuCD-F, the Escherichia coli ABC transporter for vitamin B12 uptake.Biochemistry. 2005 Dec 13;44(49):16301-9. doi: 10.1021/bi0513103. Biochemistry. 2005. PMID: 16331991
-
ABC transporter architecture and mechanism: implications from the crystal structures of BtuCD and BtuF.FEBS Lett. 2004 Apr 30;564(3):264-8. doi: 10.1016/S0014-5793(04)00289-3. FEBS Lett. 2004. PMID: 15111107 Review.
-
Structure and mechanism of ABC transporters.Curr Opin Struct Biol. 2004 Aug;14(4):426-31. doi: 10.1016/j.sbi.2004.06.005. Curr Opin Struct Biol. 2004. PMID: 15313236 Review.
Cited by
-
Energy transport pathway in proteins: Insights from non-equilibrium molecular dynamics with elastic network model.Sci Rep. 2018 Jun 22;8(1):9487. doi: 10.1038/s41598-018-27745-y. Sci Rep. 2018. PMID: 29934573 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

