Translational Control of the HIV Unspliced Genomic RNA
- PMID: 26247956
- PMCID: PMC4576183
- DOI: 10.3390/v7082822
Translational Control of the HIV Unspliced Genomic RNA
Abstract
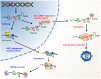
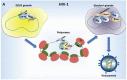
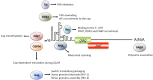
Post-transcriptional control in both HIV-1 and HIV-2 is a highly regulated process that commences in the nucleus of the host infected cell and finishes by the expression of viral proteins in the cytoplasm. Expression of the unspliced genomic RNA is particularly controlled at the level of RNA splicing, export, and translation. It appears increasingly obvious that all these steps are interconnected and they result in the building of a viral ribonucleoprotein complex (RNP) that must be efficiently translated in the cytosolic compartment. This review summarizes our knowledge about the genesis, localization, and expression of this viral RNP.
Keywords: DDX3; HIV-1; IRES; Rev; cap-dependent translation; nuclear export.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

