Boronic acid-containing aminopyridine- and aminopyrimidinecarboxamide CXCR1/2 antagonists: Optimization of aqueous solubility and oral bioavailability
- PMID: 26248802
- PMCID: PMC4564001
- DOI: 10.1016/j.bmcl.2015.07.090
Boronic acid-containing aminopyridine- and aminopyrimidinecarboxamide CXCR1/2 antagonists: Optimization of aqueous solubility and oral bioavailability
Abstract
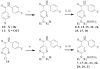
The chemokine receptors CXCR1 and CXCR2 are important pharmaceutical targets due to their key roles in inflammatory diseases and cancer progression. We have previously identified 2-[5-(4-fluoro-phenylcarbamoyl)-pyridin-2-ylsulfanylmethyl]-phenylboronic acid (SX-517) and 6-(2-boronic acid-5-trifluoromethoxy-benzylsulfanyl)-N-(4-fluoro-phenyl)-nicotinamide (SX-576) as potent non-competitive boronic acid-containing CXCR1/2 antagonists. Herein we report the synthesis and evaluation of aminopyridine and aminopyrimidine analogs of SX-517 and SX-576, identifying (2-{(benzyl)[(5-boronic acid-2-pyridyl)methyl]amino}-5-pyrimidinyl)(4-fluorophenylamino)formaldehyde as a potent chemokine antagonist with improved aqueous solubility and oral bioavailability.
Keywords: Antagonist; Asthma; COPD; CXCR1; CXCR2.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures
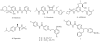
Similar articles
-
Discovery of 2-[5-(4-Fluorophenylcarbamoyl)pyridin-2-ylsulfanylmethyl]phenylboronic Acid (SX-517): Noncompetitive Boronic Acid Antagonist of CXCR1 and CXCR2.J Med Chem. 2014 Oct 23;57(20):8378-97. doi: 10.1021/jm500827t. Epub 2014 Oct 8. J Med Chem. 2014. PMID: 25254640 Free PMC article.
-
Boronic acid-containing CXCR1/2 antagonists: Optimization of metabolic stability, in vivo evaluation, and a proposed receptor binding model.Bioorg Med Chem Lett. 2015 Jun 1;25(11):2280-4. doi: 10.1016/j.bmcl.2015.04.041. Epub 2015 Apr 23. Bioorg Med Chem Lett. 2015. PMID: 25933594 Free PMC article.
-
Discovery of 2-hydroxy-N,N-dimethyl-3-{2-[[(R)-1-(5- methylfuran-2-yl)propyl]amino]-3,4-dioxocyclobut-1-enylamino}benzamide (SCH 527123): a potent, orally bioavailable CXCR2/CXCR1 receptor antagonist.J Med Chem. 2006 Dec 28;49(26):7603-6. doi: 10.1021/jm0609622. J Med Chem. 2006. PMID: 17181143
-
Combined anti CXC receptors 1 and 2 therapy is a promising anti-inflammatory treatment for respiratory diseases by reducing neutrophil migration and activation.Pulm Pharmacol Ther. 2015 Oct;34:37-45. doi: 10.1016/j.pupt.2015.08.002. Epub 2015 Aug 10. Pulm Pharmacol Ther. 2015. PMID: 26271598 Review.
-
Small molecule antagonists of the CXCR2 and CXCR1 chemokine receptors as therapeutic agents for the treatment of inflammatory diseases.Curr Top Med Chem. 2006;6(13):1345-52. doi: 10.2174/15680266106061345. Curr Top Med Chem. 2006. PMID: 16918453 Review.
Cited by
-
Discovery of 1,5-Dihydro-4H-imidazol-4-one Derivatives as Potent, Selective Antagonists of CXC Chemokine Receptor 2.ACS Med Chem Lett. 2021 Apr 8;12(5):836-845. doi: 10.1021/acsmedchemlett.1c00113. eCollection 2021 May 13. ACS Med Chem Lett. 2021. PMID: 34055234 Free PMC article.
-
The Boron Advantage: The Evolution and Diversification of Boron's Applications in Medicinal Chemistry.Pharmaceuticals (Basel). 2022 Feb 22;15(3):264. doi: 10.3390/ph15030264. Pharmaceuticals (Basel). 2022. PMID: 35337063 Free PMC article. Review.
-
Targeting Myeloid-Derived Suppressor Cell Trafficking as a Novel Immunotherapeutic Approach in Microsatellite Stable Colorectal Cancer.Cancers (Basel). 2023 Nov 20;15(22):5484. doi: 10.3390/cancers15225484. Cancers (Basel). 2023. PMID: 38001744 Free PMC article. Review.
-
Targeting CXCR2 signaling in inflammatory lung diseases: neutrophil-driven inflammation and emerging therapies.Naunyn Schmiedebergs Arch Pharmacol. 2025 Aug;398(8):9583-9607. doi: 10.1007/s00210-025-03970-x. Epub 2025 Mar 6. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 40047857 Review.
-
Role of the CXCL8-CXCR1/2 Axis in Cancer and Inflammatory Diseases.Theranostics. 2017 Apr 7;7(6):1543-1588. doi: 10.7150/thno.15625. eCollection 2017. Theranostics. 2017. PMID: 28529637 Free PMC article. Review.
References
-
- Busch-Petersen J. Curr Top Med Chem. 2006;6:1345. - PubMed
-
- Dwyer MP, Yu Y. Curr Top Med Chem. 2014;14:1590. - PubMed
-
- Boppana NB, Devarajan A, Gopal K, Barathan M, Bakar SA, Shankar EM, Ebrahim AS, Farooq SM. Exp Biol Med (Maywood) 2014;239:509. - PubMed
-
- Baggiolini M. J Intern Med. 2001;250:91. - PubMed
-
- O’Byrne PM, Naji N, Gauvreau GM. Clin Exp Allergy. 2012;42:706. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information

