Chirality Transfer in Gold(I)-Catalysed Direct Allylic Etherifications of Unactivated Alcohols: Experimental and Computational Study
- PMID: 26248980
- PMCID: PMC4586480
- DOI: 10.1002/chem.201501607
Chirality Transfer in Gold(I)-Catalysed Direct Allylic Etherifications of Unactivated Alcohols: Experimental and Computational Study
Abstract
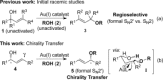
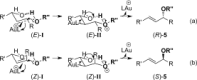
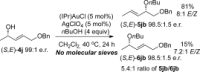
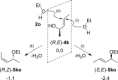
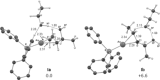
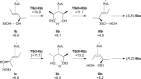
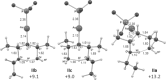
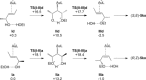
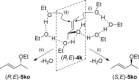
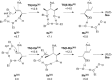
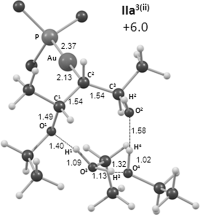
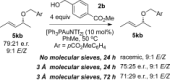
Gold(I)-catalysed direct allylic etherifications have been successfully carried out with chirality transfer to yield enantioenriched, γ-substituted secondary allylic ethers. Our investigations include a full substrate-scope screen to ascertain substituent effects on the regioselectivity, stereoselectivity and efficiency of chirality transfer, as well as control experiments to elucidate the mechanistic subtleties of the chirality-transfer process. Crucially, addition of molecular sieves was found to be necessary to ensure efficient and general chirality transfer. Computational studies suggest that the efficiency of chirality transfer is linked to the aggregation of the alcohol nucleophile around the reactive π-bound Au-allylic ether complex. With a single alcohol nucleophile, a high degree of chirality transfer is predicted. However, if three alcohols are present, alternative proton transfer chain mechanisms that erode the efficiency of chirality transfer become competitive.
Keywords: alcohols; allylations; asymmetric reactions; chirality transfer; gold.
© 2015 The Authors. Published by Wiley-VCH Verlag GmbH & Co. KGaA. This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Figures






Similar articles
-
Chirality Transfer in Gold(I)-Catalysed Hydroalkoxylation of 1,3-Disubstituted Allenes.Chemistry. 2016 Dec 19;22(51):18593-18600. doi: 10.1002/chem.201603918. Epub 2016 Nov 11. Chemistry. 2016. PMID: 27862422 Free PMC article.
-
Rhenium-catalyzed 1,3-isomerization of allylic alcohols: scope and chirality transfer.J Org Chem. 2006 Sep 29;71(20):7813-25. doi: 10.1021/jo061436l. J Org Chem. 2006. PMID: 16995691
-
Gold(I)-catalysed alcohol additions to cyclopropenes.Org Biomol Chem. 2010 Sep 21;8(18):4090-5. doi: 10.1039/c0ob00085j. Epub 2010 Jul 9. Org Biomol Chem. 2010. PMID: 20623054
-
Gold(I)-Catalysed Hydroarylation of 1,3-Disubstituted Allenes with Efficient Axial-to-Point Chirality Transfer.Chemistry. 2018 May 11;24(27):7002-7009. doi: 10.1002/chem.201800209. Epub 2018 Apr 26. Chemistry. 2018. PMID: 29542832
-
Transition metal-catalyzed allylic substitution reactions with unactivated allylic substrates.Chem Soc Rev. 2015 Nov 21;44(22):7929-67. doi: 10.1039/c5cs00144g. Epub 2015 Aug 21. Chem Soc Rev. 2015. PMID: 26293479 Review.
Cited by
-
Recent progress in the chemistry of β-aminoketones.RSC Adv. 2022 Aug 31;12(38):24681-24712. doi: 10.1039/d2ra03864a. eCollection 2022 Aug 30. RSC Adv. 2022. PMID: 36128366 Free PMC article. Review.
-
Rapid Iododeboronation with and without Gold Catalysis: Application to Radiolabelling of Arenes.Chemistry. 2018 Jan 19;24(4):937-943. doi: 10.1002/chem.201704534. Epub 2017 Dec 14. Chemistry. 2018. PMID: 29105856 Free PMC article.
-
Chirality Transfer in Gold(I)-Catalysed Hydroalkoxylation of 1,3-Disubstituted Allenes.Chemistry. 2016 Dec 19;22(51):18593-18600. doi: 10.1002/chem.201603918. Epub 2016 Nov 11. Chemistry. 2016. PMID: 27862422 Free PMC article.
References
-
- Buckingham J. Dictionary of Natural Products, Vol. 1. Cambridge, MA: University Press; 1994.
-
- For selected recent examples, see:
-
- Onitsuka K, Okuda H, Sasai H. Angew. Chem. Int. Ed. 2008;47:1454–1457. - PubMed
- Angew. Chem. 2008;120
-
- Lu Z, Ma S. Angew. Chem. Int. Ed. 2008;47:258–297. - PubMed
- Angew. Chem. 2008;120
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

