Antagonistic Interplay between MicroRNA-155 and IL-10 during Lyme Carditis and Arthritis
- PMID: 26252010
- PMCID: PMC4529177
- DOI: 10.1371/journal.pone.0135142
Antagonistic Interplay between MicroRNA-155 and IL-10 during Lyme Carditis and Arthritis
Abstract
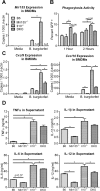
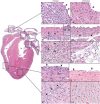
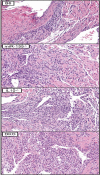
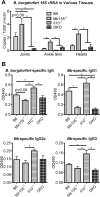
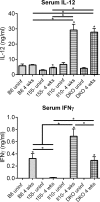
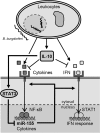
MicroRNA-155 has been shown to play a role in immune activation and inflammation, and is suppressed by IL-10, an important anti-inflammatory cytokine. The established involvement of IL-10 in the murine model of Borrelia burgdorferi-induced Lyme arthritis and carditis allowed us to assess the interplay between IL-10 and miR-155 in vivo. As reported previously, Mir155 was highly upregulated in joints from infected severely arthritic B6 Il10-/- mice, but not in mildly arthritic B6 mice. In infected hearts, Mir155 was upregulated in both strains, suggesting a role of miR-155 in Lyme carditis. Using B. burgdorferi-infected B6, Mir155-/-, Il10-/-, and Mir155-/- Il10-/- double-knockout (DKO) mice, we found that anti-inflammatory IL-10 and pro-inflammatory miR-155 have opposite and somewhat compensatory effects on myeloid cell activity, cytokine production, and antibody response. Both IL-10 and miR-155 were required for suppression of Lyme carditis. Infected Mir155-/- mice developed moderate/severe carditis, had higher B. burgdorferi numbers, and had reduced Th1 cytokine expression in hearts. In contrast, while Il10-/- and DKO mice also developed severe carditis, hearts had reduced bacterial numbers and elevated Th1 and innate cytokine expression. Surprisingly, miR-155 had little effect on Lyme arthritis. These results show that antagonistic interplay between IL-10 and miR-155 is required to balance host defense and immune activation in vivo, and this balance is particularly important for suppression of Lyme carditis. These results also highlight tissue-specific differences in Lyme arthritis and carditis pathogenesis, and reveal the importance of IL-10-mediated regulation of miR-155 in maintaining healthy immunity.
Conflict of interest statement
Figures
Similar articles
-
A joint effort: The interplay between the innate and the adaptive immune system in Lyme arthritis.Immunol Rev. 2020 Mar;294(1):63-79. doi: 10.1111/imr.12837. Epub 2020 Jan 13. Immunol Rev. 2020. PMID: 31930745 Free PMC article. Review.
-
MicroRNA-146a provides feedback regulation of lyme arthritis but not carditis during infection with Borrelia burgdorferi.PLoS Pathog. 2014 Jun 26;10(6):e1004212. doi: 10.1371/journal.ppat.1004212. eCollection 2014 Jun. PLoS Pathog. 2014. PMID: 24967703 Free PMC article.
-
Induction of Interleukin 10 by Borrelia burgdorferi Is Regulated by the Action of CD14-Dependent p38 Mitogen-Activated Protein Kinase and cAMP-Mediated Chromatin Remodeling.Infect Immun. 2018 Mar 22;86(4):e00781-17. doi: 10.1128/IAI.00781-17. Print 2018 Apr. Infect Immun. 2018. PMID: 29311239 Free PMC article.
-
Recruitment of macrophages and polymorphonuclear leukocytes in Lyme carditis.Infect Immun. 2007 Feb;75(2):613-20. doi: 10.1128/IAI.00685-06. Epub 2006 Nov 13. Infect Immun. 2007. PMID: 17101663 Free PMC article.
-
Mechanisms of Borrelia burgdorferi internalization and intracellular innate immune signaling.Front Cell Infect Microbiol. 2014 Dec 15;4:175. doi: 10.3389/fcimb.2014.00175. eCollection 2014. Front Cell Infect Microbiol. 2014. PMID: 25566512 Free PMC article. Review.
Cited by
-
A joint effort: The interplay between the innate and the adaptive immune system in Lyme arthritis.Immunol Rev. 2020 Mar;294(1):63-79. doi: 10.1111/imr.12837. Epub 2020 Jan 13. Immunol Rev. 2020. PMID: 31930745 Free PMC article. Review.
-
Spirochetal Lipoproteins and Immune Evasion.Front Immunol. 2017 Mar 29;8:364. doi: 10.3389/fimmu.2017.00364. eCollection 2017. Front Immunol. 2017. PMID: 28424696 Free PMC article. Review.
-
MicroRNA Expression Shows Inflammatory Dysregulation and Tumor-Like Proliferative Responses in Joints of Patients With Postinfectious Lyme Arthritis.Arthritis Rheumatol. 2017 May;69(5):1100-1110. doi: 10.1002/art.40039. Arthritis Rheumatol. 2017. PMID: 28076897 Free PMC article.
-
Aspects of the Immunopathogenesis of Lyme Arthritis.Microorganisms. 2025 Jul 7;13(7):1602. doi: 10.3390/microorganisms13071602. Microorganisms. 2025. PMID: 40732111 Free PMC article. Review.
-
Robust interferon signature and suppressed tissue repair gene expression in synovial tissue from patients with postinfectious, Borrelia burgdorferi-induced Lyme arthritis.Cell Microbiol. 2019 Feb;21(2):e12954. doi: 10.1111/cmi.12954. Epub 2018 Oct 17. Cell Microbiol. 2019. PMID: 30218476 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

