Cell-Size Control
- PMID: 26254313
- PMCID: PMC4744813
- DOI: 10.1101/cshperspect.a019083
Cell-Size Control
Abstract
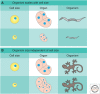
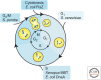
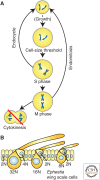
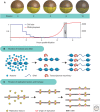
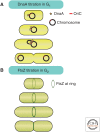
Cells of a given type maintain a characteristic cell size to function efficiently in their ecological or organismal context. They achieve this through the regulation of growth rates or by actively sensing size and coupling this signal to cell division. We focus this review on potential size-sensing mechanisms, including geometric, external cue, and titration mechanisms. Mechanisms that titrate proteins against DNA are of particular interest because they are consistent with the robust correlation of DNA content and cell size. We review the literature, which suggests that titration mechanisms may underlie cell-size sensing in Xenopus embryos, budding yeast, and Escherichia coli, whereas alternative mechanisms may function in fission yeast.
Copyright © 2016 Cold Spring Harbor Laboratory Press; all rights reserved.
Figures

References
-
- Amir A. 2014. Cell size regulation in bacteria. Phys Rev Lett 112: 208102.
-
- Baroni MD, Monti P, Alberghina L. 1994. Repression of growth-regulated G1 cyclin expression by cyclic AMP in budding yeast. Nature 371: 339–342. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
