Synergistic activity of BET protein antagonist-based combinations in mantle cell lymphoma cells sensitive or resistant to ibrutinib
- PMID: 26254443
- PMCID: PMC4582333
- DOI: 10.1182/blood-2015-04-639542
Synergistic activity of BET protein antagonist-based combinations in mantle cell lymphoma cells sensitive or resistant to ibrutinib
Erratum in
-
Sun B, Shah B, Fiskus W, et al. Synergistic activity of BET protein antagonist-based combinations in mantle cell lymphoma cells sensitive or resistant to ibrutinib. Blood. 2015;126(13):1565-1574.Blood. 2016 Sep 29;128(13):1778. doi: 10.1182/blood-2016-08-734863. Blood. 2016. PMID: 27688783 Free PMC article. No abstract available.
Abstract
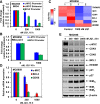
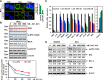
Mantle cell lymphoma (MCL) cells exhibit increased B-cell receptor and nuclear factor (NF)-κB activities. The bromodomain and extra-terminal (BET) protein bromodomain 4 is essential for the transcriptional activity of NF-κB. Here, we demonstrate that treatment with the BET protein bromodomain antagonist (BA) JQ1 attenuates MYC and cyclin-dependent kinase (CDK)4/6, inhibits the nuclear RelA levels and the expression of NF-κB target genes, including Bruton tyrosine kinase (BTK) in MCL cells. Although lowering the levels of the antiapoptotic B-cell lymphoma (BCL)2 family proteins, BA treatment induces the proapoptotic protein BIM and exerts dose-dependent lethality against cultured and primary MCL cells. Cotreatment with BA and the BTK inhibitor ibrutinib synergistically induces apoptosis of MCL cells. Compared with each agent alone, cotreatment with BA and ibrutinib markedly improved the median survival of mice engrafted with the MCL cells. BA treatment also induced apoptosis of the in vitro isolated, ibrutinib-resistant MCL cells, which overexpress CDK6, BCL2, Bcl-xL, XIAP, and AKT, but lack ibrutinib resistance-conferring BTK mutation. Cotreatment with BA and panobinostat (pan-histone deacetylase inhibitor) or palbociclib (CDK4/6 inhibitor) or ABT-199 (BCL2 antagonist) synergistically induced apoptosis of the ibrutinib-resistant MCL cells. These findings highlight and support further in vivo evaluation of the efficacy of the BA-based combinations with these agents against MCL, including ibrutinib-resistant MCL.
© 2015 by The American Society of Hematology.
Figures





Similar articles
-
Biological rational for sequential targeting of Bruton tyrosine kinase and Bcl-2 to overcome CD40-induced ABT-199 resistance in mantle cell lymphoma.Oncotarget. 2015 Apr 20;6(11):8750-9. doi: 10.18632/oncotarget.3275. Oncotarget. 2015. PMID: 25797245 Free PMC article.
-
Cell-cycle reprogramming for PI3K inhibition overrides a relapse-specific C481S BTK mutation revealed by longitudinal functional genomics in mantle cell lymphoma.Cancer Discov. 2014 Sep;4(9):1022-35. doi: 10.1158/2159-8290.CD-14-0098. Epub 2014 Jul 31. Cancer Discov. 2014. PMID: 25082755 Free PMC article.
-
BET protein proteolysis targeting chimera (PROTAC) exerts potent lethal activity against mantle cell lymphoma cells.Leukemia. 2018 Feb;32(2):343-352. doi: 10.1038/leu.2017.207. Epub 2017 Jun 30. Leukemia. 2018. PMID: 28663582
-
Ibrutinib for mantle cell lymphoma.Future Oncol. 2016 Feb;12(4):477-91. doi: 10.2217/fon.15.342. Epub 2016 Jan 13. Future Oncol. 2016. PMID: 26759179 Review.
-
Pirtobrutinib: First Non-covalent Tyrosine Kinase Inhibitor for Treating Relapsed or Refractory Mantle Cell Lymphoma in Adults.Curr Med Chem. 2024;31(30):4757-4762. doi: 10.2174/0109298673251030231004052822. Curr Med Chem. 2024. PMID: 37818564 Review.
Cited by
-
CDKN1C-mediated growth inhibition by an EZH1/2 dual inhibitor overcomes resistance of mantle cell lymphoma to ibrutinib.Cancer Sci. 2021 Jun;112(6):2314-2324. doi: 10.1111/cas.14905. Epub 2021 May 1. Cancer Sci. 2021. PMID: 33792119 Free PMC article.
-
Achieving clinical success with BET inhibitors as anti-cancer agents.Br J Cancer. 2021 Apr;124(9):1478-1490. doi: 10.1038/s41416-021-01321-0. Epub 2021 Mar 15. Br J Cancer. 2021. PMID: 33723398 Free PMC article. Review.
-
The BET bromodomain inhibitor exerts the most potent synergistic anticancer effects with quinone-containing compounds and anti-microtubule drugs.Oncotarget. 2016 Nov 29;7(48):79217-79232. doi: 10.18632/oncotarget.12640. Oncotarget. 2016. PMID: 27764794 Free PMC article.
-
Overcoming Ibrutinib Resistance in Chronic Lymphocytic Leukemia.Cancers (Basel). 2019 Nov 21;11(12):1834. doi: 10.3390/cancers11121834. Cancers (Basel). 2019. PMID: 31766355 Free PMC article. Review.
-
Combinatorial inhibition of BTK, PI3K-AKT and BRD4-MYC as a strategy for treatment of mantle cell lymphoma.Mol Biomed. 2022 Jan 15;3(1):2. doi: 10.1186/s43556-021-00066-9. Mol Biomed. 2022. PMID: 35031886 Free PMC article.
References
-
- Rossi D, Ciardullo C, Gaidano G. Genetic aberrations of signaling pathways in lymphomagenesis: revelations from next generation sequencing studies. Semin Cancer Biol. 2013;23(6):422–430. - PubMed
-
- Kridel R, Meissner B, Rogic S, et al. Whole transcriptome sequencing reveals recurrent NOTCH1 mutations in mantle cell lymphoma. Blood. 2012;119(9):1963–1971. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

