Role of Tumor Necrosis Factor Superfamily in Neuroinflammation and Autoimmunity
- PMID: 26257732
- PMCID: PMC4507150
- DOI: 10.3389/fimmu.2015.00364
Role of Tumor Necrosis Factor Superfamily in Neuroinflammation and Autoimmunity
Abstract
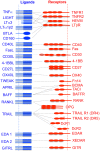
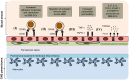
Tumor necrosis factor superfamily (TNFSF) molecules play an important role in the activation, proliferation, differentiation, and migration of immune cells into the central nervous system (CNS). Several TNF superfamily molecules are known to control alloimmunity, autoimmunity, and immunity. Development of transgenic and gene knockout animals, and monoclonal antibodies against TNFSF molecules have increased our understanding of individual receptor-ligand interactions, and their intracellular signaling during homeostasis and neuroinflammation. A strong clinical association has been observed between TNFSF members and CNS autoimmunity such as multiple sclerosis and also in its animal model experimental autoimmune encephalomyelitis. Therefore, they are promising targets for alternative therapeutic options to control autoimmunity. Although, TNFSF ligands are widely distributed and have diverse functions, we have restricted the discussions in this review to TNFSF receptor-ligand interactions and their role in the pathogenesis of neuroinflammation and CNS autoimmunity.
Keywords: autoimmunity; blood–brain barrier; multiple sclerosis; neuroinflammation; tumor necrosis factor.
Figures

References
-
- Matsumoto M. Role of TNF ligand and receptor family in the lymphoid organogenesis defined by gene targeting. J Med Invest (1999) 46:141–50. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

