Thymic Crosstalk Coordinates Medulla Organization and T-Cell Tolerance Induction
- PMID: 26257733
- PMCID: PMC4507079
- DOI: 10.3389/fimmu.2015.00365
Thymic Crosstalk Coordinates Medulla Organization and T-Cell Tolerance Induction
Abstract
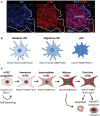
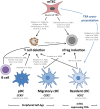
The thymus ensures the generation of a functional and highly diverse T-cell repertoire. The thymic medulla, which is mainly composed of medullary thymic epithelial cells (mTECs) and dendritic cells (DCs), provides a specialized microenvironment dedicated to the establishment of T-cell tolerance. mTECs play a privileged role in this pivotal process by their unique capacity to express a broad range of peripheral self-antigens that are presented to developing T cells. Reciprocally, developing T cells control mTEC differentiation and organization. These bidirectional interactions are commonly referred to as thymic crosstalk. This review focuses on the relative contributions of mTEC and DC subsets to the deletion of autoreactive T cells and the generation of natural regulatory T cells. We also summarize current knowledge regarding how hematopoietic cells conversely control the composition and complex three-dimensional organization of the thymic medulla.
Keywords: T-cell tolerance; autoimmune regulator; dendritic cells; medulla; medullary thymic epithelial cells; natural regulatory T cells; negative selection; thymic crosstalk.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

